Abstract
The field study was carried out on 13 rookeries in the Gulf of California in Mexico, during the breeding season of 1997 in the months of June and July. A total of 288 (137 males, 151 females) California sea lion pups (Zalophus californianus) were anesthetized for 6–25 minutes (mean=12.51, SD=4.38) with isoflurane. In a period of 20 days, the average for an island was 24 animals (range: 16–29 animals), from an estimated 2–42 days of age, weighing 6–17.4 kg (mean=10.41 kg, SD=2.02). The inhalant anesthesia with isoflurane proved to be effective and safe for the immobilization of free-ranging California sea lion pups. The morphometric measurements obtained were more precise than the ones taken when only physical restraint was used.
Introduction
The California sea lion is widely distributed in the Gulf of California in Mexico. The current population for this area is considered to be around 30,000 animals, in 40 rookeries. Thirteen of these are reproductive rookeries, harboring up to 93% of the population during the breeding season. Eighteen rookeries are non-reproductive (7%) and the rest are temporary resting places.1,7
In the last few years in Mexico, research on population dynamics, ecology and interactions of these animals with the fishing industry has intensified. The direct handling of California sea lion pups has been carried out since the 80s using physical restraint in order to mark, weigh, and measure animals. Direct observations of the quality of the breeding rookeries have suggested that there are significant differences between them.7 However, since the verification of this information requires the safe handling of animals in order to take accurate measurements and samples, chemical immobilization has been required.
As part of a long-term study on the ecology, behavior, physiology and epidemiology of the California sea lion, the use of isoflurane as an anesthetic agent for handling a great number of pups (0–6 weeks) was evaluated in several colonies from the Gulf of California. The goals were to obtain quick, quality morphometric measurements and biologic samples, and to reduce the negative impact on the breeding rookeries. In recent years, the use of the inhaled anesthesia for marine mammals has gained popularity, since it provides a quick induction, an ideal state of immobilization, a high margin of safety and a complete and quick recovery from the anesthesia.2-6
Methods
The field study was carried out on 13 rookeries in the Gulf of California, Mexico. Anesthesia was performed on 11 of them. Transportation to the islands was made possible with the help of a Mexican Navy ship that patrols the area supporting research. The Gulf of California harbors more than 100 islands, all of them with desert climate, and the great majority possess rocky beaches with difficult access, which are preferred by the California sea lions for breeding.7 The study was carried out during the breeding season of 1997 in the months of June and July. The team arrived at each island at 8:00 a.m. and departed at dusk, in order to continue on to the next rookery.
During the anesthesia, the following was obtained: Morphometric measurements (total length, curvilinear longitude, cranial, axillary, abdominal and pelvic circumference, as well as the thickness of the blubber layer at several levels); body weight; volume of the animal by means of water density immersion; hair samples to study levels of heavy metals; blood (∼10 ml) from the jugular vein for hematology and tests against infectious agents. On animals that presented cutaneous vesicles, cultures for viral isolation were collected. Fecal samples were also obtained. Samples that could not be stored were processed on the same day they were collected.
The work team consisted of three persons managing the anesthesia, taking samples and measurements, and two persons capturing, looking after the pups and returning them as required. Each animal was restrained manually and placed in a shaded plastic tub filled with water. A maximum of ten pups were kept while waiting to be handled. For the induction of the anesthesia, the animals were placed on a shaded wooden worktable. The animals were masked with a plastic cone attached to an anesthesia machine for small species (Anesco Laboratories). Isoflurane (Forane Abbott Laboratories Ltd., Queensborough. Kent, UK) was administered at 5% in oxygen (1–2 L/min) until relaxation was achieved in order to allow endotracheal intubation or sample collection. Only a few animals where intubated. In those cases, a 4–6-mm endotracheal tube (Kendall-Gammatron Ltd., UK) was used depending on the animal’s weight. The endotracheal tube was place with direct visualization with a laryngoscope. The isoflurane concentration was adjusted according to responses to stimuli and the presence or absence of palpebral reflex, and mandibular tone. The animals stayed in ventral recumbency except when moved to facilitate the taking of measurements and samples.
For the application of the anesthesia, an Isotec 3 (Ohmeda, UK) vaporizer was used, adapted to an anesthesia machine for small species with a 1-L rebreathing bag. The anesthesia equipment weighed 15 kg. Once installed in the place where the anesthesia was to take place, the isoflurane was placed in the vaporizer. The vaporizer was drained at the end of each day.
From the beginning of the anesthesia, all the physical signs were monitored. These included: palpebral reflex; capillary refill time; mandibular tone; breathing by means of the thoracic movements; and heart rate, saturation of oxygen and temperature by means of a pulse oximeter with a rectal probe. Once the animal recovered, it remained for a few minutes in a shady place before being returned to the rookery. The animal was observed from a distance.
Results
A total of 288 pups (137 males and 151 females) were anesthetized in a period of 20 days, from an estimated age ranging from 2–42 days, weighing 6–17.4 kg (mean=10.41 kg, SD=2.02). Table 1 shows the data obtained during anesthesia. In the cases of the Consagrada and Lobos islands, the pulse rate and oximetry readings are not reported. The inhalation anesthesia allowed the collection of samples and measurements from still animals, enabling the team to obtain blood samples from 80% of the individuals, compared with 40% in previous years when anesthesia was not used. The morphometric measurements were more accurate compared to the ones taken during physical restraint. It was possible to work on 16–29 animals per island (mean=24 pups), which were anesthetized for 6–25 minutes (mean=12.51, SD=4.38), in an average time of 6 hours of work time per day. The project’s schedule and goals were achieved.
Table 1. Physiologic and anesthesia parameters from 288 (137 males, 51 females) California sea lion pups in 11 islands in the Gulf of California, Mexico
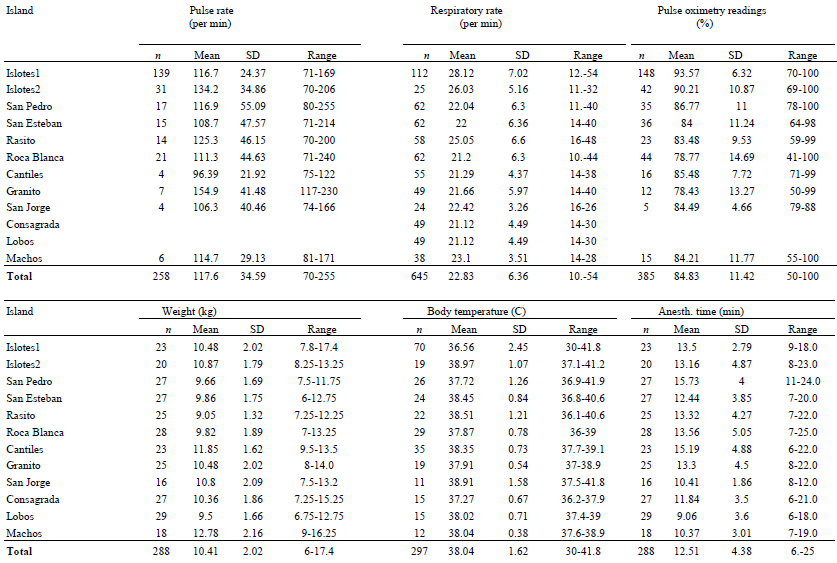
During induction, some animals were apneic for short periods, slowing the induction, and causing a marked bradycardia and a decrease in blood oxygen saturation.2 All of these animals resumed breathing and returned to the normal physiologic values without any handling. This behavior was observed in animals with a more active temperament. Four animals regurgitated milk during the anesthesia and one of them died after the anesthesia. Another animal died of hyperthermia prior to anesthesia.
Discussion
The isoflurane anesthesia proved to be effective and safe for the immobilization of free-ranging California sea lion pups and allowed us to obtain better data in a situation where work time is limited by tides and climatic conditions.
Part of the study involved estimating the nutritional status and health of the populations by evaluating the physical condition of the pups on different islands. Pup health was also reflected in differences in the average weights, difficulty of handling, difficulty of induction and also the concentration of isoflurane needed for the maintenance of the desired state of anesthesia.
The time of induction varied from 30 seconds–3 minutes, varying depending on the weight and excitement of the animals. The estimated average induction time was 1 minute. When endotracheal intubation was performed, the anesthetic planes were maintained with a lower concentration of isoflurane (1.5–2%), and better physiologic values were observed. However, the desire to handle the animals for only a short time and the type of anatomic measurements needed made it more practical to only mask the animals. But since cases of regurgitation and apneas were observed, intubation whenever possible is recommended.
The time of recovery in all animals was less than 1 minute. The pulse oximeter readings proved to be practical although not completely effective. Some readings, especially during the apneas, were as low as 41% of saturation, and the animals recovered equally fast once breathing restarted.
It was determined that of the two deaths during procedures, one (0.34%) was related to aspiration, and the other one (0.34%) was due to hyperthermia. The latter animal was not anesthetized. Due to the geographic location and the time of the year, hyperthermia is the main factor to consider before, during and after the handling of the pups. In some individuals, temperatures were up to 41.5°C. In order to control this, the animals were kept wet and in the shade at all times.4
The use of isoflurane, as compared to halothane, used in other field studies with marine mammal pups, 5,6 proved to have quicker induction and recovery times. Isoflurane allowed good control of the depth of anesthesia in the pups as reported for adults.2,3 Apnea was more frequently observed as compared to the use of halothane.6
The cost of the anesthetic equipment is justifiable, since a higher number of pups were sampled, and more accurate samples and measurements were taken. A total of 300 ml of isoflurane was used, for the 288 pups. During this project, some mature females, weighing up to 120 kg, were anesthetized for the placement of time-depth recorder devices, with excellent results. The same anesthetic equipment was used.
Acknowledgments
The authors gratefully acknowledge the entire personnel of the CICIMAR who participated in the two field trips in the Gulf of California during the breeding season of 1997, in the “Evaluation of the health status of the population of the California sea lion” project. We also thank the Armada of Mexico, especially the crew of the Gunboat C-78 Zamora, who helped carry out the project.
Literature Cited
1. Aurioles-Gamboa, D., and Zavala, G.A. 1994. Ecological factors that determine the distribution and abundance of the California sea lion (Zalophus californianus) in the Gulf of California. Ciencias Marinas. 20: 535–553.
2. Heard D. J., and D. O. Beusse. 1993. Combination detomidine, ketamine, and isoflurane anesthesia in California sea lions. (Zalophus californianus). J Zoo Wildl. Med. 24: 168–170.
3. Heath R.B., B. S. Calkins, B. S. McAllister, W. Taylor, and T. Spraker. 1996. Telazol and isoflurane field anesthesia in free-ranging Steller’s sea lions (Eumetopias jubatus). J. Zoo Wildl. Med. 27: 35–45.
4. Heath R.B., R. DeLong, V. Jameson, D. Bradley, and T Spreaker. 1997. Isoflurane anesthesia in free ranging sea lion pups. J. Wildl. Dis. 33: 206–209.
5. Ridgway S. H., and J. G. Simpson. 1969. Anesthesia and restraint for the California sea lion, Zalophus californianus. J. Am. Vet. Assoc. 155: 1059–1063.
6. Work. T. M., R. L. DeLong, T. R. Spraker, and S. R. Melin. 1993. Halothane anesthesia as a method of immobilizing free-ranging California sea lions (Zalophus californianus). J. Zoo Wildl. Med. 24:482–487.
7. Zavala, G.A. 1990. La poblaci—n del lobo marino comœn, Zalophus californianus (Lesson, 1828) en las Islas del Golfo de California, Mexico. Tesis de Licenciatura. Facultad de Ciencias. UNAM. 253 Pp.