Department of Animal Reproduction and Veterinary Radiology, School of Veterinary Medicine and Animal Science, UNESP, Botucatu, São Paulo, Brazil
Abstract
Vaginal smears from nine capybaras (Hydrochoerus hydrochaeris) were examined at 3-day intervals for 30 days in order to describe the cyclic patterns of epithelial cells exfoliated through this period. The vaginal cells were classified as basal, parabasal, small intermediate, medium intermediate, large intermediate, superficial, and anucleate and their dimensions were measured to feature each one. The maturation index (MI) and karyopyknotic indexes (KI) were determined for each specimen and then compared with plasmatic estradiol to progesterone (E2/P4) ratio. The median plasmatic values and pattern deviations observed from estradiol and progesterone were respectively, 326.49 ± 202.61 pg/ml and 121.61 ± 59.17 ng/ml, and the intra-assay coefficient of variation was 11.01%.
Introduction
Vaginal cytology can serve as a crude but reliable indirect estrogen assay.4 Cytologic changes on vaginal epithelium through the estrous cycle of capybara have been described.2 As the vaginal wall epithelium starts to thicken the cells move further from the blood supply and die. Therefore, the different vaginal cell types represent stages of cell death. The basal and parabasal cells are the healthiest cells of vaginal epithelium, they are round and the nucleus is big. As they move further from the blood supply, the cells become larger and more irregular in shape, while the nucleus becomes progressively smaller, then pyknotic, before disintegrating.4 The basic principle of hormonal evaluation by means of vaginal cytology is based on the assumption that the degree of cellular maturity is related to the actual amount of sex steroids present. In certain conditions the degree of maturity of the vaginal epithelium may be proportional to the degree of the estrogenic effect. Hormonal evaluations are sometimes expressed by means of indices; however, these are meaningful only when they relate to a previous index on the same specimen.3 There are several different indices and the two most significant are the maturation index (MI) and the karyopyknotic index (KI). The maturation index is the percentage of parabasal, intermediate, and superficial cells (P:I:S) and the karyopyknotic index is the ratio of superficial cells to intermediate cells. These indexes have not been previously described for capybaras.
There are few reports regarding hormonal characteristics of capybara. The patterns of progesterone were described in serum,2 ranging from 0.9–3.5 ng/ml, and on feces where 3.52 ng/g was considered the threshold between follicular and luteal phases (unpublished data). No estradiol values were described. Hematologic evaluation may also be used as a parameter to determine animal stress, specifically the presence of leukocytosis and neutrophilia.5 Normal hematologic values for capybara have been described.1
The purpose of this study was to examine and describe exfoliated vaginal cells in the capybara and to determine if plasmatic estradiol to progesterone (E2/P4) ratio was correlated with the maturation index (MI) and the karyopyknotic index (KI).
Methods
Vaginal smears from nine healthy capybaras of Parque do Sabiá Zoo (Uberlândia/MG, Brazil) were examined at 3-day intervals for 30 days. The animals were sedated using ketamine (15 mg/kg) and midazolam (0.2 mg/kg) combination applied with darts. The cells were collected from the lateral wall of vagina with a smooth sterile brush and the smears immediately fixed in an alcohol-ether solution (1:1). The fixed smears were stained with Shorr and 100 cells were counted in ocular microscopy at 10X and 40X magnification. The vaginal cells were classified as basal, parabasal, small intermediate, medium intermediate, large intermediate, superficial, and anucleate in accordance with cytoplasm acidity and dimensional measurements made with a micrometric ocular Zeiss Kpl W/OX coupled on a 100X magnifier. The higher size of each cell was considered the length and its perpendicular line (90°) was the width (Fig. 1). The same procedure was repeated for cell nuclei. The nucleus to cytoplasm (N/C) ratios was determined using the area of cells calculated by length times width, as if the cells were rectangular. The MI and the KI were determined for each specimen by forming three subgroups: parabasal cells (formed by basal and parabasal cells), intermediate cells (formed by small, medium, and large intermediate cells), and superficial cells (formed by superficial and anucleate cells). MI equal or higher than 70% was determined to characterize an estrous animal. We collected blood from four animals to measure hematologic alterations that can be caused by stress.
| Figure 1 | 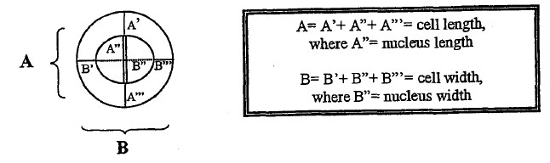
Schematic cell measurement. |
|
| |
To assess the plasmatic levels of estradiol and progesterone, blood was collected from the cephalic or femoral veins with heparinized vacutainer tubes. Samples were refrigerated until centrifuged at 800 g for 10 minutes. Separated plasma was kept frozen at -20°C until assayed. Estradiol and progesterone were assayed using solid-phase radioimmunoassay (RIA) kits without previous extraction (Coat-A-Count Kit, Diagnostic Products Company, Los Angeles, CA). All samples were assayed in a single assay to avoid inter-assay variation. The plasmatic hormonal levels were compared with the MI and KI.
Results
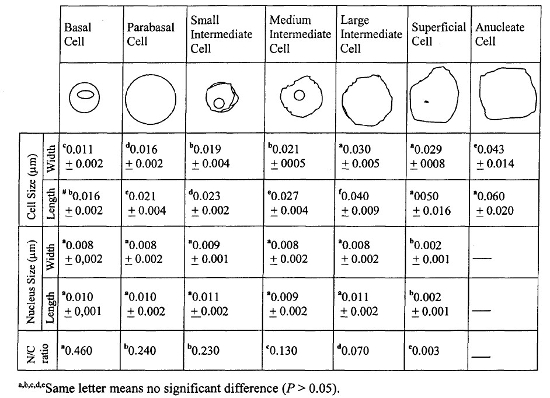
The cytoplasm acidity, cellular morphology and dimensions, and the nucleus to cytoplasm ratio characterized all cell types (Table 1). We identified all seven cells types of vaginal epithelium in capybara and, as the cells grew in size, acidity increased and the cytoplasmic color changed from blue-green to red-brown. The smallest cell was the basal, followed by parabasal, small intermediate, medium intermediate, large intermediate, superficial, and anucleate, in crescent order, as in other mammals. While cells grew, the nucleus diminished as the nucleus to cytoplasm ratio decreased. The MI and KI were calculated for each specimen.
Table 1. Vaginal epithelial cells characterization by dimensions (median ± SD) of cytoplasm and nucleus, and the nucleus to cytoplasm ratio
The minimum and maximum detection limit of the assays were, respectively, 15.51–4335.31 pg/ml for estradiol and 0.10–41.72 ng/ml for progesterone. The medium plasmatic values and pattern deviations observed from estradiol and progesterone were, respectively, 326.49 ± 202.61 pg/ml and 121.61 ± 59.17 ng/ml. Intra-assay coefficient of variation was 11.01%. The E2/P4 ratio values were plotted with MI and KI indexes for each animal (Fig. 2).
| Figure 2 | 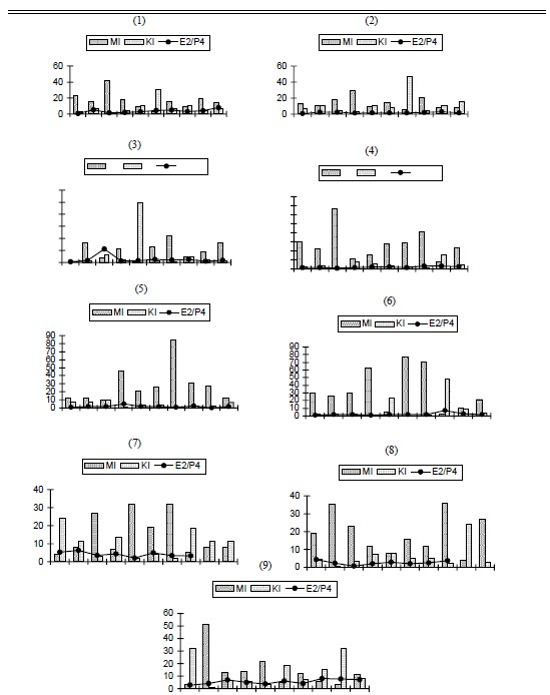
E2/P4 ratio and MI and KI index relationship, animals 1–9 (each bar graph represents one animal). |
|
| |
The linear association between estradiol and progesterone did not exist. To the regression equation y=127.3 + 1.08x (y = estradiol, and x = progesterone) the correlation was 0.459 with p=0.0636 (non-significant). We did not find a positive correlation between high estradiol levels and the vaginal cytology with 70% or more superficial cells, as we expected. Hematologic values did not reveal a stress development and ranged between the normal accepted values.
Discussion
The vaginal cells of capybara can be characterized as similar to the vaginal epithelial cells from other mammals. With cellular death some organelles can break and release acid contents, so that the cytoplasm of a superficial cell is more acidic than that of a basal cell. This justifies the different colors for different cells with the same stains methods. The cytologic changes on vaginal epithelium through the estrous cycle of capybara was not observed, as previously described.2 The MI and KI were not assessed before and we considered those as the first reference. The patterns of progesterone found in plasma did not match with the values described in serum or feces. There were no estradiol values previously described to capybara that can be used to compare our data. The absence of a positive correlation between the high estradiol levels and vaginal cytology was not well understood but we suspect that three things interfered with our data: (1) the chemicals used to sedate the animal probably changed the reproductive patterns; (2) even without hematologic changes, we suspected that restraint stressed the animals and this can also change the reproductive patterns; and (3) we do not know if the RIA kits used to evaluate hormonal levels are compatible with capybara. This paper can be used as a first reference. More attention should be given to this species as it is a prolific breeder in captivity (like many other rodents) and because capybara are raised as a farm animal in many countries of South America.
Acknowledgments
We would like to thank FAPESP for financial support.
Literature Cited
1. Arouca ME, Miranda LB, Lopes RS, Takahira RK, Kohayagawa A, Ciarlini PC, Oba E. Valores hematológicos de capivaras (Hydrochoerus hydrochaeris) criadas em cativeiro no município de Botucatu - SP. Ciência Rural - UFSM. 2000;30(5).
2. Barbella SL. Determinación del ciclo estral en chigüire (Hydrochoerus hydrochaeris). Acta Científica Venezolana. 1982;33:497–501.
3. Bibbo M, Wied G. Hormonal Cytology. In: Comprehensive Cytopathology. Philadelphia, PA: W.B. Saunders; 1991:85–114.
4. Feldman EC, Nelson RW. Ovarian cycle and vaginal cytology. In: Canine and Feline Endocrinology and Reproduction. Philadelphia, PA: W.B. Saunders; 1996:526–546.
5. Fowler ME. Stress. In: Zoo and Wild Animal Medicine. Philadelphia, PA: W.B. Saunders; 1986:35.