Abstract
Urinary estrogen, pregnanediol, estrone, estradiol, and androsterone values during pregnancy and menstrual cycles of orangutans have been studied by several investigators.4-7,9 Serum values for estradiol, progesterone, testosterone and luteinizing hormone (LH) have been evaluated during the menstrual cycles of orangutans and macaques.10,12
In this ongoing study, serum progesterone, β-human chorionic gonadotropin (β-HCG) and estradiol-17β were serially monitored before, during and after three successive pregnancies in an adult female Bornean orangutan (Pongo pygmaeus pygmaeus). Results were compared to a nonpregnant 33-yr-old female Bornean orangutan in the same facility, and human reference normals. Blood samples were obtained during weekly to biweekly conditioning programs without sedatives or tranquilizers. Transabdominal ultrasound images were obtained during the later stages of the first pregnancy, and throughout gestation in subsequent pregnancies. Differences in β-HCG and progesterone values resulting in a stillborn infant were retrospectively observed to differ from two subsequent successful births.
Serum progesterone (4-prenen-3,20-dione), β-HCG, and estradiol [1,3,5 (10)-estratrien-3,17 β-diol] values were determined using microparticle enzyme immunoassays (AxSYM®, Abbott Laboratories, Abbott Park, IL, USA) performed on samples obtained from the orangutan through a stationary conditioning program. These samples were frozen at -86°C and analyzed at a later date.
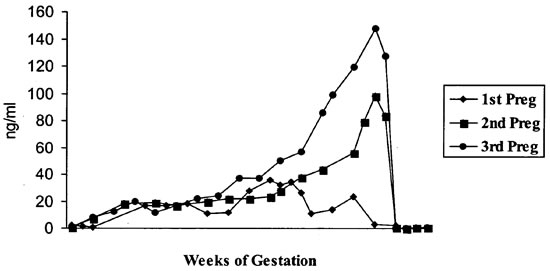
Values for serum progesterone, β-HCG, and estradiol are presented in Figures 1-3. Results of pregestational, gestational and postgestational values closely parallel human normals.2,13,14 However, significant differences were noted in serum progesterone and β-HCG values of the first pregnancy compared to two subsequent successful pregnancies. In humans, progesterone is produced primarily by the corpus luteum of the ovary.1 Progesterone prepares the uterus for implantation and maintains the uterus during pregnancy. During the menstrual cycle in humans, serum progesterone values elevate to 10–20 ng/ml 5–7 days after ovulation. During the sixth week of pregnancy, the placenta becomes the major source of progesterone.8 Values during this time elevate from 10–50 ng/ml during the first trimester to 50–2,809 ng/ml during the third trimester.2,13 In all three pregnancies, progesterone levels were between 10–15 ng/ml during the first trimester, and elevated to 30–40 ng/ml during the second trimester. However, progesterone levels decreased to 10–15 ng/ml during the third trimester of the first pregnancy (Figure 1). Serum levels during this pregnancy began to elevate to previous values but never fully recovered. Reasons for this are unknown, but we speculate that these values indicated the potential for gestational complications. Although this pregnancy proceeded to term, the infant was stillborn. An underlying cause was not determined.
Figure 1. Serum progesterone values (ng/ml)
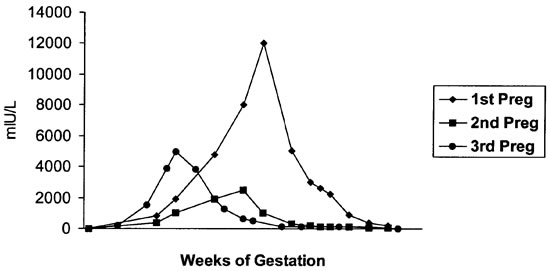
Beta-HCG appears to maintain the corpus luteum during early pregnancy in humans, allowing continued secretion of progesterone, which supports the endometrium. Values for serum β-HCG in humans demonstrate a maximal peak in the late first trimester, with a decline to a fairly constant level by mid-gestation.2 In our study, serum β-HCG values during the first pregnancy elevated beyond the values for the subsequent pregnancies and remained elevated for an extended period when compared to the two subsequent gestations. We speculate that this value maintained elevated levels due to fetal complications of excreting progesterone to maintain a healthy pregnancy (Figure 2).
Figure 2. Serum β-HCG values (mIU/L)
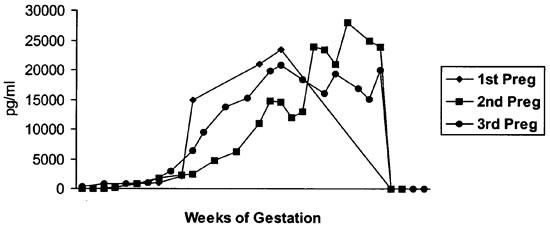
Estradiol levels in human studies demonstrate a constant rise until parturition.11,14 Levels of 100–500 pg/ml occur during the first trimester, 5,000–15,000 during the second trimester, and 10,000–40,000 pg/ml during the third trimester.14 A similar curve was observed in this group of data for all three pregnancies (Figure 3).
Figure 3. Serum estradiol values (pg/ml)
In this orangutan, values for serum progesterone and β-HCG may have predicted gestational complications resulting in a stillborn infant. Two subsequent pregnancy evaluations demonstrated parallel values and both resulted in uncomplicated births. These values may provide a basis for evaluating serum reproductive hormone levels in Bornean orangutans during pregnancy.
Acknowledgments
The authors would like to thank the orangutan care staff for the long-term conditioning of the orangutans making this study possible.
Literature Cited
1. Abraham G.E., W.D. Odell. 1972. Simultaneous radioimmunoassay of plasma FSH, LH, progesterone, 17 hydroxyprogesterone, and estradiol-17β during the menstrual cycle. J. Clin. Endocrinol. 34:312–318.
2. Braunstein G.D., J. Rasor, D. Adler, H. Danzer, and M.E. Wade. 1976. Serum human chorionic gonadotropin in normal and pathologic pregnancy. J. Clin. Endocrinol. Metab. 126:678–681.
3. Buster, J.E. and G.E. Abraham. 1975. The application of steroid hormone radioimmunoassays to clinical obstetrics. Obstet. Gynecol. 46(4):489–499.
4. Collins, D.C., C.E. Graham, and J.R.K. Preedy. 1975. Identification and measurement of urinary estrone, estradiol-17β, estriol, pregnanediol and androsterone during the menstrual cycle of the orangutan. Endo. 96(1):93–101.
5. Czekala N.M., K. Benirschke, H. McClure, and B.L. Lasley. 1983. Urinary estrogen excretion during pregnancy in the gorilla (Gorilla gorilla), orangutan (Pongo pygmaeus) and the human (Homo sapiens). Biol. Repro. 28:289–294.
6. DeGroot, L.J.(ed). Endocrinology. 2nd edition, Vol 3, Ovarian Hormone Synthesis, Circulation, and Mechanism of Action. W.B. Saunders Co, Philadelphia, 1989. Pp. 2048–2050.
7. Graham, C.E. 1988. Reproductive physiology. In: Schwarts J.H. (ed.) Orangutan Biology. Oxford University Press, New York, Pp.91–101.
8. Hertig, A.T. and R.B. Livingston. 1944. Spontaneous, threatened and habitual abortion; their pathogenesis and treatment. N. Engl. J. Med. 230:797–805.
9. Lasley, B.L., J.K. Hodges, and N.M. Czekala. 1980. Monitoring the female reproductive cycle of great apes and other primate species by determination of oestrogen and LH in small volumes of urine. J. Reprod. Fert. 28:121–129.
10. Nadler, R.D., D.C. Collins, and M.S. Blank. 1984. Luteinizing hormone and gonadal steroid levels during the menstrual cycle of orangutans. J. Med. Primatol. 3:305–314.
11. Ryan, K.J. 1980. Placental synthesis of steroid hormones. In: Tulchinksy, D., K.J. Ryan (eds.) Maternal-Fetal Endocrinology. W.B. Saunders Co, Philadelphia, Pp. 3–16.
12. Shideler, S.E., C.J. Munro, L. Tell, G. Owiti, L. Laughlin, R. Chatterton, and B.L. Lasley. 1990. The relationship of serum estradiol and progesterone concentrations to the enzyme immunoassay measurements of urinary estrone conjugates and immunoreactive prenanediol-3-glucuronide in Macaca mulatta. Am. J. Primatol. 22:113–122.
13. Speroff, L., R.H. Glass, and N.G. Kase. 1994. The endocrinology of pregnancy. In: Mitchell, C. (ed.). Clinical Gynecologic Endocrinology and Infertility. 5th edition. Williams and Wilkins Pp. 251–289.
14. Winkel, P., P. Gaede, and J. Lyngbye. 1976. Method for monitoring plasma progesterone concentrations in pregnancy. Clin. Chem. 22:422–428.