Evaluation of an Implanted Osmotic Pump for Delivery of Amikacin in Corn Snakes (Elaphe guttata guttata)
Abstract
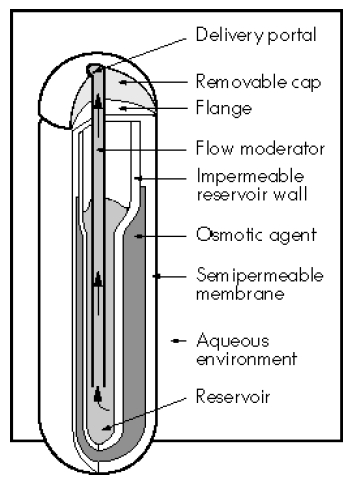
Medical treatment of venomous or aggressive snakes is a challenge for the reptile veterinarian and a safe, effective and reliable method of drug delivery for these snakes is needed. Alzet® osmotic pumps (Durect Corp., Cupertino, CA, USA, www.alzet.com) are miniature cylindric implants that operate on the basis of an osmotic pressure difference between the extracellular fluid and the osmotic agent in the pump (Figure 1). As water diffuses across the outer semipermeable membrane, the ‘salt sleeve’ compartment expands and presses on an internal flexible reservoir, thereby releasing the drug at a controlled, continuous rate. As the pump rate is defined by the semipermeable membrane and osmotic gradient, the resulting delivery rate is independent of the drug being dispensed.
Figure 1. Cutaway section of osmotic pump
Nine adult corn snakes (Elaphe guttata guttata) were used in this study. Five snakes (group A) had an osmotic pump (Alzet® Model 2002) surgically placed into the caudal coelomic cavity. The pumps were loaded with amikacin (Phoenix Scientific, Inc., St. Joseph, MO, USA) at a concentration designed to deliver 0.026 mg/kg/h and each snake was administered a loading dose of 1.69 mg/kg of amikacin intramuscularly at the time of implantation. Blood samples were obtained via cardiocentesis at 0, 2, 24, 48, 72, 144, 216, and 288 h after pump placement. Pumps were removed after 288 h and blood samples obtained 48 and 96 h after removal. Four snakes (group B) received intramuscular injections of amikacin every 72 h at a dose of 5 mg/kg for the first injection and 2.5 mg/kg for each additional injection for a total of 4 injections. Blood samples were obtained just prior to and 2 h after each injection, and at 72 h after the fourth injection. Samples were analyzed for amikacin via by fluorescence polarization immunoassay (TDx®, Abbott Laboratories, Abbott Park, IL, USA). Renal function was evaluated before and after the study via serum uric acid and phosphorus concentrations, and via renal scintigraphy with 99mTc-mercaptoacetyltriglycine (99mTc-MAG3).
The target serum concentration of amikacin was 8 µg/ml. The mean serum concentration for group A was 6.31 µg/ml (SD 0.86). The mean pump rate was determined to be the same as the expected rate of 0.13 µl/h (SD 0.017, range: 0.12–0.16). Mean peak serum concentration for group B was 16.1 µg/ml (SD 2.7) and mean trough concentrations were 8.1 µg/ml (SD 1.1). Mean serum concentration for group A was less than the mean trough concentration in group B. No evidence of decreased renal function was detected via serum chemistry analysis or renal scintigraphy in either group. One snake died due to migration of the pump into the trachea after inadvertent placement of the pump into the caudal air sac.
The Alzet® osmotic pumps were highly reliable and efficacious in delivering amikacin to corn snakes at a predictable and steady rate. They were easy to load, place and remove. Due to the potential for migration within the coelomic cavity, intracoelomic placement may not be appropriate for some snake-pump size combinations. Additional studies are planned to evaluate subcutaneous placement of these pumps in corn snakes.