Abstract
In April 2004, a 12-year-old male cheetah (Acinonyx jubatus) presented with polydipsia and moderate weight loss. The cheetah had an unremarkable health history and had been normoglycemic for the past 10 years. Following presentation, a blood sample taken under restraint in a squeeze cage showed hyperglycemia (357 mg/dl, ISIS 132±35 mg/dl). Subsequent samples revealed persistent hyperglycemia although an initial fructosamine value of 362 µmol/L was within normal limits for domestic carnivores.1 Concomitant glucosuria on free-catch urine was also noted. A diagnosis of diabetes mellitus was made.
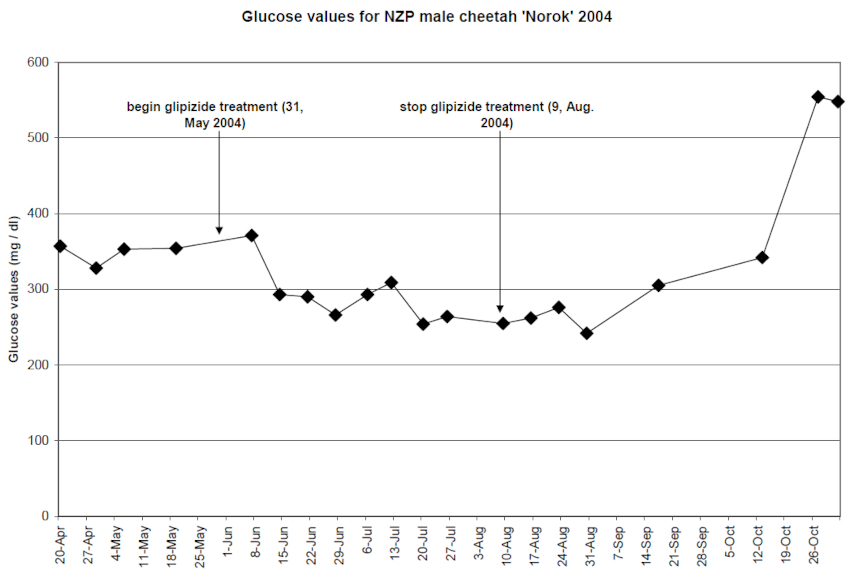
Treatment with the oral hypoglycemic glipizide (Glipizide tablets, Watson Laboratories, Inc., Corona, CA, USA) was started at a conservative dose of 2.5 mg PO BID (Figure 1). As side effects were not noted, the dose was increased to 5.0 mg on day 10. Response to treatment was monitored with weekly blood and free-catch urine samples. Treatment with glipizide reduced blood glucose levels for over 70 days (from 371–255 mg/dl). However, this therapy had to be discontinued due to severe elevation of liver enzymes believed to be caused by the drug (Figure 1).
Figure 1. Glucose values of a 12-year-old male cheetah
Insulin therapy was next considered. However, blood samples revealed elevated serum cortisol levels (11 µg/dl, ISIS 6.7±0.2 µg/dl). In addition, urine analysis showed an elevated cortisol:creatinine (C:Cr) ratio of 7.3 (healthy cheetah: 1.1; domestic cat: <1.0). Concern arose that insulin resistance caused by the hyperadrenal state would complicate insulin therapy. Ultrasound revealed moderate bilateral hyperplasia of the adrenal glands but no evidence of adrenal tumor (AT). Adrenal hyperplasia is a common finding in captive cheetahs.2 Ultrasound also showed multiple nodular areas throughout the pancreas.
Further diagnostics were performed to determine the cause of hypercortisolemia. A blood sample was taken to determine endogenous ACTH and baseline cortisol levels. Results showed that endogenous ACTH was similar to the value of a non-diabetic cheetah (26.8 pg/ml and 34.6 pg/ml, respectively) and considered non-diagnostic for either pituitary-dependent hyperadrenocorticism (PDH) or adrenocortical neoplasia. A high-dose dexamethasone suppression test (HDDST) was also performed. Dexamethasone sodium phosphate (0.1 mg/kg) was injected intravenously in the lateral tail vein using a 23-G butterfly while the cheetah was gently restrained in a squeeze cage. A subsequent blood sample was collected at 8 hours from dexamethasone injection using the same technique described above. The HDDST showed a suppression of the plasma cortisol concentration from 11.1–3.19 µg/dl at 8 hours. In domestic animals, an 8-hour post-dexamethasone plasma cortisol concentration that is less than 50% of the baseline concentration is suggestive of PDH.1 Adrenal tumor was ruled out. A new urine analysis showed a persistently elevated C:Cr ratio (3.4) further supporting the diagnosis of hyperadrenocorticism. However, an MRI of the head using 7 ml of gadolinium (0.5 mmol/ml) IV to enhance the pituitary gland revealed no pituitary abnormalities. MRI of the abdomen confirmed moderate hyperplasia of the adrenal glands and the presence of multiple cluster-like nodules scattered through the pancreas.
Insulin therapy was initiated with 6 then 8 units of ultralente insulin, (Humulin U [Ultralente], Eli Lilly and Co., Indianapolis, IN, USA) SC SID. Unfortunately, within a week the cheetah developed severe azotemia (BUN 145 mg/dl, ISIS 38±12 mg/dl), creatinine 9.1 mg/dl (ISIS 2.7±0.8 mg/dl) and exacerbated hyperglycemia (glucose >500 mg/dl). The cheetah was euthanatized at this time.
Gross necropsy revealed a moderately enlarged pancreas thickened by multiple cysts containing clear fluid or white, mucoid material. Histologically, cysts were solitary or multiloculated and lined by cuboidal epithelium. Dilated ducts often contained thick, proteinaceous material. There were multiple areas of interstitial fibrosis, and cells of the islets of Langerhans were frequently displaced by amyloid-like material.
Literature Cited
1. Nelson, R.W., and C.G. Couto. Disorders of the adrenal gland. In: Small Animal Internal Medicine. 3rd Edition. Nelson, R.W., and C.G. Couto (eds.). Mosby. St. Louis, Missouri. 2003.
2. Terio, K., L. Marker, and L. Munson. 2004. Evidence for chronic stress in captive but not free-ranging cheetahs (Acinonyx jubatus) based on adrenal morphology and function. J Wildl Dis. 40(2):259–266.