Introduction
Acute kidney injury (AKI) and chronic kidney disease (CKD) are common in dogs and cats and are associated with substantial morbidity and mortality. Serum creatinine is the most commonly utilized marker for renal function, but is neither sensitive nor specific for early detection of kidney injury.
Limitations of Serum Creatinine Concentration
Despite the diagnostic advancements made in other medical fields (e.g., the use of biomarkers in cardiology), serum creatinine is still being used as the marker for kidney function despite its multiple shortcomings.
The main limitation of serum creatinine include the following:
1. Serum creatinine is affected by extra-renal factors, like hydration status and muscle mass, therefore it lacks specificity. Creatinine may be increased in dehydrated animals in face of normal kidney function. In cachectic animals (common in animals with advanced CKD) creatinine concentration will overestimate kidney function.
2. Creatinine is influenced by muscle mass; therefore, its baseline concentration varies among dog breeds. The attempt to include all dogs under one reference range results in a very wide reference range, which decreases its sensitivity.
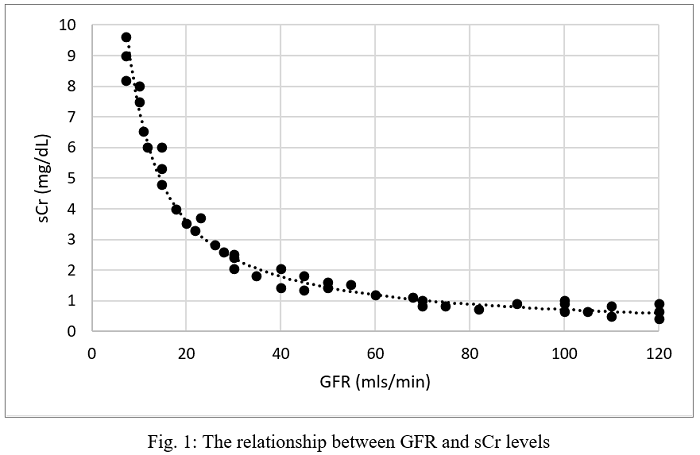
3. The nonlinear relationship between glomerular filtration rate (GFR) and creatinine (Fig. 1) also de- creases its sensitivity since substantial changes in GFR at the early stages of the disease, are associated with only small changes in serum creatinine concentration, thus creatinine will not increase above its reference range until substantial loss in GFR has already occurred. This phenomenon is more pronounced in dogs with low baseline creatinine concentration (dogs with low muscle mass). In most dog breeds creatinine is not expected to rise above the reference range until ~75% of nephrons become non-functional.
4. Serum creatinine is a functional marker thus it is “blinded” to kidney injury that is not accompanied by decreased kidney function. In the liver, different markers are indicative to liver injury compared to liver function. Increased activities of alanine aminotransferase for example indicate liver injury but are not indicative of liver function. Other markers (i.e., urea, glucose, cholesterol, albumin, bilirubin, bile acids, etc.) indicate presence of liver failure. Thus the clinician is aware of the presence of liver injury and can intervene before failure ensues. Conversely, in the kidney, there are no readily available and sensitive markers of kidney injury, thus with the first indication of kidney disease, substantial decrease in kidney function has already occurred.
5. Serum creatinine does not represent the severity of the dysfunction until a steady state has been reached. For example, if GFR drops to zero and serum creatinine is measured immediately, it will still be within the reference range. In time, serum creatinine will increase but it will take days until steady state has been reached, and the severity of the injury can be accurately assessed. Consequently, substantial changes in GFR at the early stages of AKI are associated with relatively small changes in serum creatinine.
The above limitations of serum creatinine are being reflected by the findings of several studies in human medicine indicating that small, and even transient, increase in serum creatinine concentration in human patients is detrimental. In one study, as little as 0.5 mg/dL increase in serum creatinine was associated with increased in-hospital mortality1. In another study, a transient increase in serum creatinine was also associated with increased odds ratio for in-hospital mortality2. Finally, even a small and transient increase in serum creatinine in patients that were discharged from the hospital, was associated with the need for chronic dialysis over the ensuing three years 3. In a study of heatstroke in dogs, the median creatinine concentration was only mildly increased at presentation and third of the dogs presented with normal serum creatinine concentration4; however, concurrent evaluation of kidney function using GFR measurement revealed that kidney function was markedly decreased. Moreover, when kidney injury was assessed using sensitive markers, presence of kidney injury was identified in 100% of the dogs4.
The aforementioned limitations and studies imply that relying on serum creatinine as the only marker of kidney function does not provide the entire information needed to accurately assess kidney function. When relying on serum creatinine, AKI is characteristically recognized only when the disease is within the maintenance phase, when clinical signs are overt. This is one of the speculated reasons for the high mortality rate among animals with AKI, as late recognition of the disease provides only a narrow window of opportunity for therapy. In CKD, due to the wide reference range, the nonlinear relationship between GFR and serum creatinine and because the disease is initially subclinical, the diagnosis is delayed until most of nephrons are already lost.
Improving the Interpretation of Serum Creatinine
Understating renal physiology and the limitations of serum creatinine improves interpretation of serum creatinine concentration. As opposed to the common believe that serum creatinine does not increase until 75% of GFR is decreased, any decrease in GFR is expected in be reflected by an increase in serum creatinine concentration. Thus, following small trends in serum creatinine concentration within the reference range increases its sensitivity. In fact, the International Renal Interest Society defines Grade I AKI as 0.3 mg/dL (26.4 µmol/L) increase in creatinine concentration compared to the baseline, even when creatinine is within the reference range. The same can be applied to animals with CKD. Once the baseline serum creatinine concentration is a known, 0.3 mg/dL increase in the same dog and under the same conditions (fasted sample, normal hydration status, same methodology) represent decrease in kidney function and presence of CKD, even if creatinine is still within the reference range. The normal baseline creatinine concentration for a specific dog should be determined in a relatively young age, when the dog is fasted and is well hydrated. Any subsequent measurements of creatinine should be related to the baseline concentration and not for the reference range of dogs.
One of the aforementioned limitations of serum creatinine relates to its variability among dog breeds5. Therefore, increased awareness for the breed specific reference range can sensitize the clinician to elevated creatinine even if the latter is still within the reference range. For example, creatinine of 1.2 in a Yorkshire Terrier likely represents ~75% decrease in kidney function, despite the fact that it is well within the reference range of most laboratories. Conversely, a serum creatinine concentration of 1.5 mg/dL in a dog with high muscle mass might be normal for the dog even of this value is above the reference range.
Serum creatinine should be interpreted with additional markers of kidney function. Blood urea nitrogen is a non-specific marker of kidney function, since it is influenced by multiple non renal parameters (e.g., dietary protein, gastrointestinal bleeding, catabolism, polyuria and polydipsia, dehydration, liver function), yet the urea to creatinine ratio should always be evaluated. For example, when the urea/creatinine ratio is increased, a potential reason might be decreased body mass, thus, in this case, creatinine is likely underrepresents the decrease in kidney function. Another marker that can be used in conjunction to serum creatinine, is symmetric dimethylarginine (SDMA). SDMA is a methylated form of the amino acid arginine and is primarily eliminated through the kidneys by renal filtration and excretion6; therefore, it is a potential endogenous marker of GFR. SDMA is superior to creatinine since is it not influenced by muscle mass; thus, these two markers should be used in conjunction.
Summary
Serum creatinine has multiple limitations, however, understanding renal physiology and its limitations improves its interpretation. There is a need for sensitive renal biomarkers that will facilitate the diagnosis of kidney diseases and will allow earlier intervention. Renal biomarkers that can identify kidney injury even in the absence of decreased kidney function will likely become available in the near future and have the potential to aid in the diagnosis of AKI and CKD.
References
1. Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol. 2005 Nov;16(11):3365–70.
2. Uchino S, Bellomo R, Bagshaw SM, Goldsmith D. Transient azotaemia is associated with a high risk of death in hospitalized patients. Nephrol Dial Transplant. 2010 Jan;25(6):1833–9.
3. Wald R, Quinn RR, Luo J, Li P, Scales DC, Mamdani MM, et al. Chronic dialysis and death among survivors of acute kidney injury requiring dialysis. JAMA. 2009 Sep;302(11):1179–85.
4. Segev G, Daminet S, Meyer E, De Loor J, Cohen A, Aroch I, et al. Characterization of kidney damage using several renal biomarkers in dogs with naturally occurring heatstroke. Vet J. 2015 Nov;206(2):231–5.
5. Braun JP, Lefebvre HP, Watson AD. Creatinine in the dog: a review. Veterinary Clinical Pathology. 2003;32(4):162–79.
6. McDermott J. Studies on the catabolism of Ng-methylarginine, Ng, Ng-dimethylarginine and Ng, Ng-dimethylarginine in the rabbit. Biochemical Journal. 1976 Jan;154(1):179–84.