M. Burrows
Division of Veterinary and Biomedical Science, Murdoch University, Murdoch, WA, Australia
Colonisation and Diversity
Malassezia pachydermatis is a non-mycelial yeast. It is approximately 2x4 micrometers in size, has an oval shape with a thick wall and reproduces by unipolar budding. This gives the organism its characteristic shape. There are at least 13 species of lipid-dependent yeast including M. sympodialis, M. equina, M. caprae, M. cuniculi (colonising animal skin) and the rest: M. furfur, M. globosa, M. obtusa, M. restricta, M. slooffiae, M. dermatis, M. japonica and M. yamatoensis colonising human skin.
Malassezia pachydermatis is present as a commensal of the skin and mucosae of most dogs. In clinically normal dogs, the sites most frequently colonised by M. pachydermatis are the ear canals, anus, lips, chin, interdigital skin, rectum and vagina. It is uncommon on the axilla, groin or dorsum. The anus seems to be the most frequently colonised mucosal site.
Pathogenesis: Dogs
It is now clear that under certain circumstances, Malassezia can change from being a commensal organism to a significant pathogen. For this to occur, there has to be alteration in the skin microclimate that allows the organism to proliferate to excessive levels. These changes may involve increased temperature and humidity, changes in skin lipids, alteration in epidermal barrier function and the presence of concurrent bacterial infection. Since S. pseudintermedius and M. pachydermatis are inhabitants of the mucosae, including the oral cavity, they will continually be transferred to the skin, particularly in areas which require cleaning or grooming, and which are pruritic. Thus there is potential for the establishment of microbial overgrowth whenever the skin is damaged or there is underlying disease impairing cutaneous function. These abnormalities may occur in association with:
- Allergic skin diseases
- Epidermal abnormalities (hepatocutaneous syndrome, zinc-responsive dermatoses)
- Endocrine diseases (hyperadrenocorticoidism, hypothyroidism)
- Breed-associated susceptibility (basset hound, dachshund, cocker spaniel, West Highland white terrier, miniature poodle, German shepherd, Cavalier King Charles spaniel are at higher risk)
- Prior treatment with antibiotics: suggested but not corroborated. Unlike Candida spp., Malassezia is not inhibited by bacteria but is enhanced by the increased growth of S. pseudintermedius.
Clinical Features: Dogs
Malassezia pachydermatis most commonly causes a pruritic dermatitis that can affect the lips, muzzle, periocular region, ventral neck, axillae, interdigital areas, body folds, medial thighs and perineum. In the ear canals, Malassezia causes an erythematous, ceruminous otitis. Depending on the sites affected, Malassezia dermatitis may present as foot chewing, frantic face rubbing, ear scratching, perianal licking or scooting or more generalised pruritus.
Commonly Affected Areas
It is important to note that all these sites may not be affected in an individual dog. Also in severely affected dogs, the lesions can become more generalised. The lesions seen in these areas consist of erythema, often covered by a yellow or slate-grey waxy scale. The skin may feel greasy to touch and there may be an offensive, rancid odour. With time, the lesions may become hyperpigmented or develop into erythematous plaques. Chronic lesions are often lichenified. Pruritus may be quite marked. Malassezia overgrowth can also be detected in the claws of some dogs resulting in a reddish-brown staining of the proximal claw or a waxy exudate in the claw fold, with inflammation of the surrounding soft tissue. In dogs, Malassezia paronychia is commonly associated with atopic dermatitis and paw licking.
Hypersensitivity
There is good evidence to support the role of M. pachydermatis as an allergen in canine atopic dermatitis. This situation, known as Malassezia hypersensitivity, can occur alongside reactivity to other allergens or it can occur in isolation. Clinically, Malassezia hypersensitivity in dogs manifests as a highly inflammatory and pruritic response mounted to relatively low numbers of yeast organisms, though some dogs may also have overt infection with cytologically evident overgrowth. Clinical diagnosis of Malassezia hypersensitivity in the dog relies upon intradermal allergy testing, or by measuring serum allergen-specific IgE. Recent studies have demonstrated good correlation between IDAT reactivity and allergen-specific IgE in atopic dogs. In humans, the clinical implications of a positive IgE result remains controversial, but more recent studies demonstrate a positive correlation between disease severity (of AD) and Malassezia-specific IgE titres. This has not yet been demonstrated in dogs. A therapeutic trial is warranted to determine the significance of the cytologic findings and results in marked improvement with resolution of pruritus. Immunotherapy for Malassezia hypersensitivity is available although published studies are limited.
Diagnosis
The diagnosis of Malassezia dermatitis is suggested by the typical history of regional pruritus and the presence of appropriate lesions in typical sites. A definitive diagnosis can be made by demonstrating excessive numbers of Malassezia organisms on the skin surface or in the ears and observing a response to specific therapy. The most useful and readily available tool for the diagnosis of Malassezia dermatitis or otitis is cytology.
Samples may be collected from the skin surface by a variety of methods, including direct impression smears, tape impression smears and dry cotton swabs, but the easiest to use is clear adhesive tape. Tape is preferred because the organisms are sometimes not located at the surface of the lesions and repeated application of the tape to the same site will reveal deeper populations. The technique is quick and easy to perform and, with experience, tapes can be examined in the microscope and diagnosis made rapidly. The presence of numbers of Malassezia greater than 2 per high power x1000 oil immersion field is suggestive of microbial overgrowth.
Commonly populations are very much higher but the organisms may be found in clusters, so at least 20 high-power fields should be examined. For diagnosis of Malassezia paronychia, the broken end of a wooden cotton-tip swab can be used to scrape the claw fold, and exudate is pressed and rolled onto a glass slide.
For examination of ear exudate in dogs and cats with ceruminous or exudative otitis externa, rolling of exudate in a thin layer on glass slides with a cotton-tip swab is the preferred method.
Treatment and Control
Topical Therapy
Malassezia dermatitis responds to topical therapy with antimicrobial shampoo agents containing chlorhexidine and miconazole (Malaseb®), piroctone olamine (Mediderm®) or econazole (Sebazole®).
It is critical that the owner is given adequate instructions when prescribing these products. They must be thoroughly massaged into the affected areas, including between the toes and in the axillae. The shampoo must be left in contact with the skin for 10 minutes before rinsing. Initially the shampoo should be used three times a week, until the condition has responded. At that time, repeat tape strips can be assessed to monitor the improvement. If a good response is seen, the frequency of bathing can be reduced to twice and then once weekly. If the underlying problem is identified and controlled, then it may not be necessary to continue long-term bathing. However if an underlying cause cannot be found, routine maintenance baths given one or twice a week can be used to keep the condition under control.
Localised areas of Malassezia dermatitis such as on the feet or lips can be treated with antifungal creams containing 2% miconazole (Daktarin®) cream, 1% clotrimazole (Canesten®) cream or lotion), nystatin or terbinafine (Lamisil®) or antifungal wipes containing 2% acetic acid, 2% boric acid q 12h or climbazole 0.5%/chlorhexidine 3% pads q 24h (Biohex®).
Malassezia otitis usually responds well to treatment with otic drops containing clotrimazole (Otomax®, Mometamax®), miconazole (Surolan®), terbinafine (Osurnia®) or nystatin (Canaural®); however, it is very important to identify an underlying cause to prevent recurrence.
Systemic Therapy
Systemic therapy for Malassezia infections may be required for severe cases or for those in which regular topical therapy is not practicable.
Ketoconazole, itraconazole, fluconazole and terbinafine have all been reported effective. Ketoconazole used to be treatment of choice but this is no longer commercially available in Australia and has to be compounded.
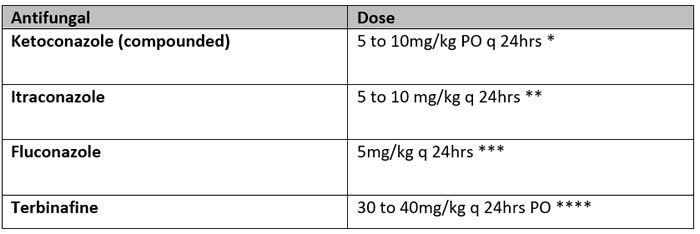
*A low-dose regimen for large dogs using ketoconazole 5 mg/kg q 12h PO for 10 days, followed by 5 mg/kg q 24h PO for 10 doses has been reported to be successful in the majority of cases, and lessens the expense of therapy.
** Itraconazole persists in the stratum corneum and therefore pulse therapy can be used. Dogs treated with 5 mg/kg q 24h for two consecutive days followed by 5 days without treatment for 3 cycles (3 weeks) responded as well as dogs who had received the medication at 5 mg/kg/day for 21 days (Pinchbeck 2002).
*** Anecdotal evidence suggests that fluconazole is not as clinically effective as the other azoles.
****Terbinafine given to dogs with dermatitis at 30 mg/kg once daily for 3 weeks resulted in a similar improvement in cytological and skin lesion scores as in dogs given the drug at the same dose twice weekly for 3 weeks; the improvement in pruritus was higher with the daily treatment.
Severe claw fold infections may require longer treatment or higher doses, however, and otitis externa cases may not respond adequately.
There are no veterinary products licensed for the treatment of Malassezia dermatitis in cats. Systemic therapy is the treatment of choice. Itraconazole is preferred to ketoconazole because it is better tolerated in cats. Fluconazole has also been used to treat Malassezia dermatitis in cats and may be a more affordable treatment option.
Prophylaxis for Chronic/Relapsing Malassezia Dermatitis
- Regular shampoo therapy (weekly or biweekly)
- Pulse oral itraconazole or terbinafine: two consecutive days each week
- Monitor for hepatotoxicity with CBC and biochemistry every 6 months
Immunotherapy for Malassezia Dermatitis
A commercial Malassezia extract is licensed and available (Greer Laboratories®). A good response to subcutaneous immunotherapy administered for a minimum of 10 months was reported in nine of 16 cases (56%) of Malassezia hypersensitivity confirmed by intradermal allergy testing with both a reduction in use of anti-inflammatory and antifungal medication as well as a reduction in pruritus scores by >50%. No adverse effects were reported (Aberg 2017).
References
1. Aberg L, Varjonen K, Ahman S. Results of allergen specific immunotherapy in atopic dogs with Malassezia hypersensitivity: a retrospective study of 16 cases. Vet Dermatol. 2017;28:633–636.
2. Bond R, et al. Factors associated with elevated cutaneous Malassezia pachydermatis populations in dogs with pruritic skin disease. J Small Anim Pract. 1996;37:103–107.
3. Chen T, Hill PB. Review: the biology of Malassezia organisms and their ability to induce immune responses and skin disease. Vet Dermatol. 2005;16:4–26.
4. Glatz M, Buchner M, von Bartenwerffer W, et al. Malassezia spp.-specific immunoglobulin E level as a marker for severity of atopic dermatitis in adults. Acta Derm Venereol. 2015;95:191–196.
5. Harada K, Saito M, Sugita T, Tsuboi R. Malassezia species and their associated skin diseases. J Dermatol. 2015;42:250–257.
6. Negre A, Bensignor E, Guillot J. Evidence-based veterinary dermatology: a systematic review of interventions for Malassezia dermatitis in dogs. Vet Dermatol. 2009;20(1):1–12.
7. Oldenhoff WE, Frank GR, DeBoer DJ. Comparison of the results of intradermal test reactivity and serum allergen-specific IgE measurements for Malassezia pachydermatis in atopic dogs. Vet Dermatol. 2014;25:507–511.