Abstract
The aim of this study was to describe the normal electrocardiographic patterns in Russell’s viper (Daboia russelii) and Indian spectacled cobra (Naja naja). Ten adult rescued vipers and ten adult rescued cobras were used for this study. The snake was restrained without anaesthesia with the help of a restraining tube and held in dorsoventral recumbency by two assistants. Traditional forelimb leads were connected two heart lengths cranial to the heart on the left side and rear limb leads were connected two heart-lengths caudal to the heart on the right side, which yielded an ECG tracing similar to standard lead II as in case of dogs. The waveforms were analyzed in lead II at 25mm/sec at 10 mm = 1 mV to determine heart rate, rhythm, P, PR, QRS, ST and QT durations and P, net QRS complex and T amplitude. The polarity of each wave was tabulated in lead II. The P waves and QRS complex were predominantly positive in standard bipolar lead II. T waves were found to be both positive and negative. Establishment of normal electrocardiogram values will help to identify abnormal ECG patterns during procedures like anesthesia and for the future investigations of cardiovascular diseases.
Introduction
Conservation of reptiles in general is of negligible importance in India since olden days. Snakes have always been known for their natural defenses. Hence the first reaction of any human being when they encounter snakes is to kill them, except a few of which are of mythological importance. Also, research undertaken in this class of the taxa in India is very minimal, thereby making the availability of diagnostic tools minimal. For successful conservation practices, understanding the biology and physiology, availability of diagnostic tools, and treatment and management of diseases are important factors. Standardization of electrocardiographic parameters may serve as a valuable diagnostic tool for various research investigations and surgical procedures.
Electrocardiography is useful for assessing cardiac rhythm and chamber size and for diagnosing conduction abnormalities.1 The electrocardiogram (ECG) can provide relevant information about electrical activity and cardiac remodeling; however, abnormal parameters suggest, but do not identify structural cardiac diseases. The ECG is a useful tool for diagnosing most arrhythmias and can provide information on the myocardial status because the deflections and intervals recorded are often altered by either pathologic or physiologic factors.2,3 It can also be used as an indicator of cardiac chamber enlargement and electrolyte imbalances.3
The ECG can be an invaluable tool for monitoring cardiac function in reptiles. Unfortunately, this diagnostic test is used sparingly by many veterinarians because of limited understanding regarding its interpretation. Because the heart rate is tied to environmental temperature, it is important to maintain a reptile within its preferred optimal temperature range when performing this test. The reptilian ECG has many of the same characteristics of a mammalian ECG.4 The present study was undertaken to establish the normal electrocardiographic parameters using a safe physical restraint. The ECG was recorded without using any anesthetic agents for the snake. Thus, the cardiac activity remained uninfluenced by chemical immobilization, and values closer to the physiologic values could be recorded.
Materials and Methods
Ten rescued adult Russell’s vipers and ten rescued Indian spectacled cobras were selected for the study. All the snakes used for the study were rescued from populated areas in and around Bangalore. Necessary permissions for acquisition were procured from the Principal Chief Conservator of Forest (Wildlife), Karnataka Forest Department. As the snakes would have undergone significant stress during the rescue activities, they were housed for two days to allow the animals to stabilize and the stressors on them to reduce. During this period, the snakes were observed for their behavior and movements. Those with normal behavior and activities were selected for the study.
The snakes were restrained with the help of standard snake hooks and restraining tubes (Midwest®). Post-restraint, a thorough physical examination was carried out for each specimen. Snakes with any injuries or physical abnormalities were not selected for the study. Five out of ten Russell’s vipers were females and five were males. Five out of ten Indian spectacled cobras were males and five were females. The length and weight of each individual snake is tabulated in Table 1.
Recording of ECG
Electrocardiograms (BPL Cardiart 9108, 2011 with simultaneous 12-channel ECG recording and 12-lead simultaneous acquisitions) of all 20 snakes were recorded under manual restraint without usage of any anesthetic agents. All snakes were held in dorsoventral recumbency on a wooden table for ECG. The tracing was recorded at a paper speed of 25 mm/second at the sensitivity of 1 with the voltage of 1 mV per 10 mm. ECGs were recorded in standard bipolar limb leads (I, II, III), and augmented unipolar limb leads (aVR, aVL, aVF); however, for calculation of various parameters, standard lead II was considered. The electrodes were attached to the skin using alligator clips after application of coupling gel (BPL Cardijelly®) liberally. The gel applied was more than the amount applied while recording ECG of mammals. Larger quantities of gel were used to attain excellent contact, as the skin of snakes is keratinized and hard. The traditional leads were placed approximately two heart-lengths cranial and caudal to the heart.4,5 The cranial leads included RA (right arm) followed by LA (left arm) and the caudal leads included RL (right leg) followed by LL (left leg). The cranial leads were placed on the left lateral side of the snake where the dorsal scales and ventral scales meet, while the caudal leads were placed on the right lateral side of the snake5 (Figure 1).
Results
Typical ECG tracings of snakes in the standard bipolar limb leads and augmented unipolar limb leads are shown in Figures 1 and 2. The mean heart rate was determined to be 66.64±10.89 beats per minute (BPM) in Russell’s vipers and 68.48±10.72 BPM in spectacled cobras.
The various ECG parameters measured in standard bipolar lead (lead II), viz. amplitude and duration of P, QRS and T waves; and the PQ, PR, ST and QT segments of each individual snake are summarized in Table 2a (Russell’s viper) and 2b (Indian spectacled cobra). The bottom two rows of the table comprise the mean and standard deviation for each particular parameter.
The QRS complexes of Indian spectacled cobra were of lesser amplitude in comparison with the QRS complexes of Russell’s viper. The mean value of the QRS amplitude in Russell’s viper was 1.27 mV whereas in Indian spectacled cobra it was 0.82 mV. The maximum recording in viper was 1.55 mV while in cobras the R wave did not exceed 0.9 mV. Other parameters of the two species did not show much difference. There were no evident differences in the ECG of males and females within the species.
In one Russell’s viper, a phenomenon similar to sinus arrhythmia in mammals was noted wherein clusters of QRS complexes were evident with extended periods of time gap between them. An approximate time duration of 2.16 seconds was recorded between the last T wave of one cluster and first P wave of the consecutive next wave. No electrical activity was recorded on the ECG during this time gap.
Discussion
The interpretation of ECG of snakes can be difficult because of limited reference material available presently for comparison. In addition, the ECG can be influenced by various environmental factors. For example, the heart rate is dependent on the body temperature, and the intervals such as the P–R and Q–T segments are influenced by the heart rate. To reduce the likelihood of misclassifying the results of an ECG, it is important to perform these tests under optimal conditions.4 In the present study the ECG was recorded with the environmental temperature within the POTZ. However, there are no established POTZ values for both the species. As both these species are generally found in tropical countries,6,7 the temperature range maintained during this study was 26–28°C. Hence, the variations in ECG that could be attributed to temperature were reduced.
Reptiles have a tremendous capacity for anaerobic metabolism and can hold their breath for extended periods of time8. This was more commonly observed in Russell’s viper than Indian spectacled cobra during this study. As seen in one of the snakes, the phenomenon similar to sinus arrhythmia could be because of prolonged breath holding. Absence of any electrical activity was witnessed during this period.
The various ECG parameters recorded in the study were found to be almost similar to the ECG recordings of canines.3 One exception seen in this study was prolonged ST interval which may be suggestive of slower depolarization of ventricle in these species.
The anatomical location of the heart in snakes is usually in the first one-third to one-fourth of the body.4,5,9 In the present study it was found that the Russell’s viper heart is located almost midway through the body. In contrast, the spectacled cobra heart was located just after the base of the hood.
To standardize the normal ECG in these species, a larger sample size must be taken into consideration so that individual variations and human errors can be nullified. There is scope for further research to be done before ECG for snakes can be standardized any further.
Table 1. Length, weight, and sex of each snake
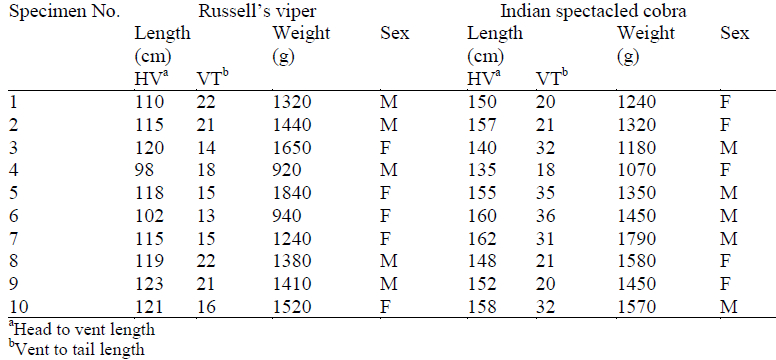
Table 2a. ECG parameters of Russell’s viper with mean and standard deviation
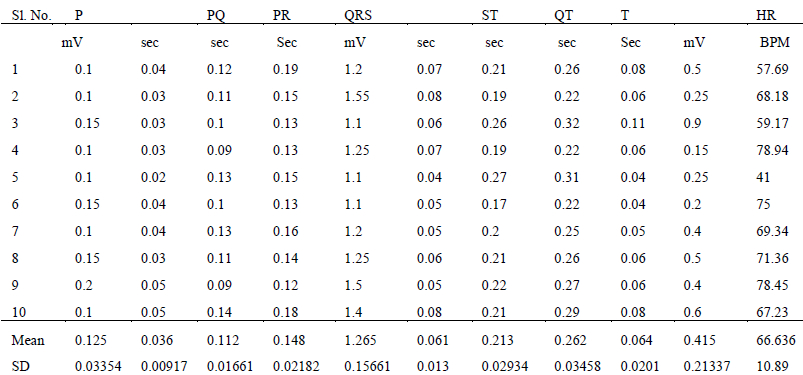
Table 2b. ECG parameters of Indian spectacled cobra with mean and standard deviation
Figure 1. Attachment of standard bipolar limb leads

Image reproduced with permission from M. Karthik.
Figure 2. ECG recording from Indian spectacled cobra showing the bipolar limb leads and augmented unipolar limb leads
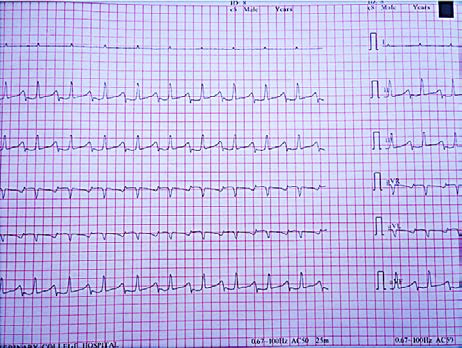
Image reproduced with permission from M. Karthik.
Figure 3. ECG recording from Russell’s viper showing the bipolar limb leads and augmented unipolar limb leads
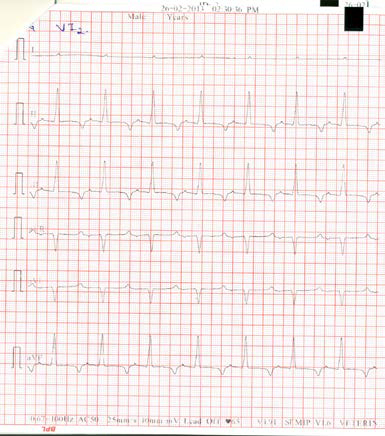
*Note the negative T waves. Image reproduced with permission from M. Karthik.
Figure 4. Magnified image of lead II of Russell’s viper
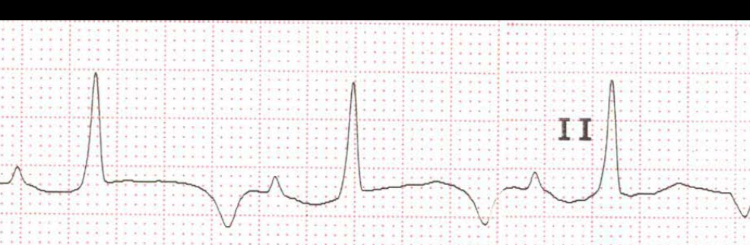
Image reproduced with permission from M. Karthik.
Literature Cited
1. Smith FWK, Hadlock DJ. Electrocardiography. In: Miller MS, Tilley LP, eds. Manual of Canine and Feline Cardiology. Philadelphia, PA: WB Saunders Co.; 1995:47–74.
2. Loypetjara P. Electrocardiography of the wildlife animals. Wetchasan Sattawaophaet. Thai J Vet Med. 1991;21:175–186.
3. Tilley LP, ed. Essentials of Canine and Feline Electrocardiography. 3rd ed. Philadelphia, PA: Lea and Febiger; 1992.
4. Marja JLK, Mark AM. Reptile cardiology: a review of anatomy and physiology, diagnostic approaches, and clinical disease. Semin Avian Exot Pet Med. 2005;14(1):52–60.
5. Murray MJ. Cardiology and circulation. In: Mader DR, ed. Reptile Medicine and Surgery. Toronto, Canada: WB Saunders; 1996:95–103.
6. Whitaker R, Captain A. Snakes of India: The Field Guide. Chennai, India: Draco Books; 2004a:304.
7. Whitaker R, Captain A. Snakes of India: The Field Guide. Chennai, India: Draco Books; 2004b:332.
8. Sladky KK, Mans C. Clinical anaesthesia in reptiles. J Exotic Pet Med. 2012;21:17–31.
9. Mosley CA. Anesthesia and analgesia in reptiles. Semin Avian Exot Pet Med. 2005;14:243–262.