Abstract
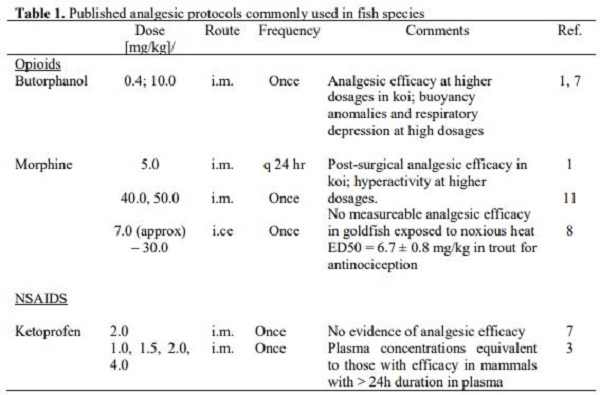
The objective of this presentation is to describe and highlight what is currently known about pain (nociception) and analgesia (antinociception) in fish. The primary question regarding fish analgesia is whether fish “experience” pain or are fish species merely capable of demonstrating a “reflexive” response to a noxious stimulus (nociception)? Of critical importance is the concept of whether we can recognize pain in fish, and is the perception of pain by a fish equivalent to that of a mammal? We will never be able to fully and objectively answer these questions, because fish simply cannot tell us. Many would argue that fish do not have the same anatomic and/or physiologic capabilities to “process” pain.13 In other words, fish are merely responding and passively reacting to stimuli to which they are exposed, with little or no ability for cognition or self-awareness.13 However, recent research has demonstrated that the transmission of peripheral sensory signals, via the spinal cord, to midbrain and forebrain regions that are homologous to mammalian cortical and limbic structures.2,5,7,15-17 Additionally, the endogenous opioid system, which is activated in response to nociception and contributes to analgesia, is also well conserved throughout vertebrate phylogeny.4,6,15 Thus, the physiologic and anatomic requirements for pain and analgesia appear to be remarkably similar among all vertebrate species, and therefore, there is substantive and compelling evidence from the neuroanatomic, neurophysiologic and behavioral literature to suggest that, at some level, a variety of fish species experience pain under certain contexts. In my clinical experience, both kappa- and mu-opioid agonists appear to be affective in providing pain relief, particularly post-surgically; however, mu-opioid agonists appear to provide fewer deleterious side effects.1,8-10,12
Many veterinary clinicians argue that the administration of analgesics is risky to the patient and may mask behavioral signs of pain, which are considered evolutionarily adaptive for survival. However, veterinarians have an ethical obligation to treat painful conditions in all animals, including fish, as effective pain management reduces stress-induced disruption to homeostatic mechanisms, and also decreases morbidity and mortality associated with trauma or surgery. Several obstacles limit successful analgesic use in fish, including subjectivity in pain assessment, inadequate knowledge of analgesic efficacy across species, pharmacokinetics of analgesic drugs, and the unknown relationship between risks and benefits for specific drugs. It is my hope that future research will help us to determine if fish feel pain. Until then, we must use all available evidence, especially in those species most closely related to the species being studied, to err on the side of animal in subjectively assessing that a procedure considered painful in a mammal, should also be considered potentially painful in a fish species.
Literature Cited
1. Baker T, Baker B, Johnson SM, Sladky KK. Comparative analgesic efficacy of morphine and butorphanol in koi (Cyprinus carpio) undergoing gonadectomy. JAVMA (in press).
2. Braithwaite VA, Boulcott P. Pain perception, aversion and fear in fish. Dis Aquat Organ. 75:131–138, 2007.
3. Davis, MR, Mylniczenko N, Storms T, Raymond F, Dunn LJ. Evaluation of intramuscular ketoprofen and butorphanol as analgesics in chain dogfish (Scyliorhinus retifer). Zoo Biol. 25:491–500, 2006.
4. de Velasco EM, Law PY, Rodríguez RE. Mu opioid receptor from the zebrafish exhibits functional characteristics as those of mammalian mu opioid receptor. Zebrafish. 6:259–268, 2009.
5. Dunlop R, Laming P. Mechanoreceptive and nociceptive responses in the central nervous system of goldfish (Carassius auratus) and trout (Oncorhynchus mykiss). J Pain. 6:561–568, 2005.
6. Gonzalez-Nunez V, Rodríguez RE. The zebrafish: a model to study the endogenous mechanisms of pain. ILAR J. 50:373–386, 2009.
7. Harms CA, Lewbart GA, Swanson CR, Kishimori JM, Boylan SM. Behavioral and clinical pathology changes in koi carp (Cyprinus carpio) subjected to anesthesia and surgery with and without intra-operative analgesics. Comp Med. 55:221–226, 2005.
8. Jones SG, Kamunde C, Lemke K, Stevens ED. The dose-response relation for the antinociceptive effect of morphine in a fish, rainbow trout. J Vet Pharmacol Ther. 2012 Jan 9. doi: 10.1111/j.1365-2885.2011.01363.x. [Epub ahead of print]
9. Newby NC, Mendonça PC, Gamperl K, Stevens ED. Pharmacokinetics of morphine in fish: winter flounder (Pseudopleuronectes americanus) and seawater-acclimated rainbow trout (Oncorhynchus mykiss). Comp Biochem Physiol C Toxicol Pharmacol. 143:275–283, 2006.
10. Newby NC, Mendonça PC, Gamperl K, Stevens ED. Pharmacokinetics of morphine in fish: winter flounder (Pseudopleuronectes americanus) and seawater-acclimated rainbow trout (Oncorhynchus mykiss). Comp Biochem Physiol C Toxicol Pharmacol. 143:275–283, 2006.
11. Nordgreen J, Garner JP, Janczak AM, Ranheim B, Muir WM, Horsberg TE. Thermonociception in fish: effects of two different doses of morphine on thermal threshold and post-test behaviour in goldfish (Carassius auratus). Appl Anim Behav Sci. 119:101–107, 2009.
12. Nordgreen J, Kolsrud HH, Ranheim B, Horsberg TE. Pharmacokinetics of morphine after intramuscular injection in common goldfish (Carassius auratus) and Atlantic salmon (Salmo salar). Dis Aquat Organ. 88:55–63, 2009.
13. Rose JD. Anthropomorphism and 'mental welfare' of fishes. Dis Aquat Organ. 75:139–154, 2007.
14. Sladky KK. I’ll have the fish and shrimps: analgesia in fish and invertebrates. www.royalsociety.org.nz/organisation/panels/anzccart/2011.
15. Proceedings, ANZCCART Annual Conference, Rotorua, New Zealand, June 26–28, 2011.
16. Sneddon LU. Evolution of nociception in vertebrates: comparative analysis of lower vertebrates. Brain Res Brain Res Rev. 46:123–130, 2004.
17. Sneddon LU. Pain perception in fish: indicators and endpoints. ILAR J. 50:338–42, 2009.
18. Weber ES. Fish analgesia: pain, stress, fear aversion, or nociception? Vet Clin North Am Exot Anim Pract. 14:21–32, 2011.