Faculdade de Medicina Veterinária e Zootecnia Universidade Estadual Paulista (UNESP) Botucatu, SP, Brazil
Introduction
Hypotension, defined as mean arterial pressure (MAP) < 60 mm Hg or systolic arterial pressure (SAP) < 90 mm Hg, is one of the most common intra-operative complications observed in small animal anesthesia. In high risk patients, such as dogs with gastric dilation and volvulus, hypotension detected at any time during the hospitalization period is one of the factors that are associated with increased risk of death.1 Higher mortality rates have been reported in critically ill cats presenting with hypotension (SAP < 90 mm Hg) during the hospitalization period.2
How to Diagnose Hypotension
The systematic use of blood pressure monitors should be encouraged. With the exception of teaching hospitals and referral centers, it seems that blood pressure monitors are not a common place in many veterinary practices, even in developed countries.3 Direct measurement of blood pressure via an arterial catheter connected to a blood pressure transducer system, is the preferred method in critically ill patients or in those that are at risk for developing major blood losses and/or hypotension during surgery. For most anesthetic procedures indirect blood pressure monitoring monitors can be used. In anesthetized dogs, non-invasive oscillometric and Doppler ultrasound monitors are effective for diagnosing hypotension.4,5 In cats and in small dogs (<5-10 kg) oscillometric devices developed for human use may not work properly. Systolic arterial pressure measured by the Doppler ultrasound underestimates SAP in cats: in one study Doppler ultrasound SAP values were closer to the MAP than to the SAP values displayed by the direct blood pressure monitor.6
Managing the Anesthetic Technique to Minimize Peri-Operative Hypotension
Mean arterial pressure is the driving pressure of blood through the tissues; this variable is determined by cardiac output (CO) and by systemic vascular resistance (SVR), according to the formula:
MAP = CO X SVR
Complex disease states and anesthesia can alter arterial pressure via changes in CO and/or in SVR. Acepromazine, a long acting tranquilizer commonly used in small animal practice, may contribute to hypotension in critically ill patients because of its blocking action on alfa-1 adrenergic receptors leading vasodilation (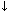 SVR). Although acepromazine can be used in healthy dogs, this drug is best avoided in animals that are presented with cardiovascular instability/hypovolemia because it will impair the vasopressor action of drugs used to treat hypotension, such as dopamine and ephedrine.7-8
SVR). Although acepromazine can be used in healthy dogs, this drug is best avoided in animals that are presented with cardiovascular instability/hypovolemia because it will impair the vasopressor action of drugs used to treat hypotension, such as dopamine and ephedrine.7-8
Inhalant anesthetics induce dose-dependent decreases in arterial blood pressure. Halothane decreases arterial blood pressure mainly via decreases in myocardial contractility and in CO; isoflurane and sevoflurane induced hypotension is caused mainly by vasodilation. In healthy dogs, anesthesia can be maintained with an inhalational anesthetic alone. However in patients that are presented for major surgery with signs of systemic illness, are at greater risk for developing intra-operative hypotension. In this population, hypotension can be minimized by reducing the dose of inhalational anesthetic by the concomitant use of constant rate infusions of opioids and other intravenous drugs in a balanced anesthesia technique.9-10 When high doses of opioids (e.g., fentanyl) during anesthesia, an improvement in arterial pressure may not become evident until antagonism of opioid induced bradycardia is achieved by the use of an anticholinergic agent.9
Blockade of sympathetic ganglia induced by local anesthetics epidurally can decrease SVR, further aggravating pre-existing hypotension. Extensive regional blocks, such as epidural anesthesia should be avoided in hypovolemic/hypotensive patients.
Intraoperative Hypotension--Therapeutic Options
Anticholinergics: Treatment of vagally mediated bradycardia (heart rate [HR] < 60-70 beats/min in dogs or < 100 beats/min in cats) with an anticholinergic agent (atropine: 0.02-0.04 mg/kg/IV, glycopyrrolate: 5-10 mcg/kg/IV) should be considered if low heart rates are associated with hypotension.11 Volume replacement therapy: Many animals are presented for surgery with variable degrees of hypovolemia. Decreased circulating volume reduces venous return leading to decreased CO and arterial pressure. Signs of hypovolemic shock (including hypotension) may be caused by extensive blood losses (> 30% of circulating volume) or severe dehydration (> 10-12% of body weight). Animals presenting with mild to moderate degrees of hypovolemia may not show signs of physiologic decompensation (e.g., hypotension) because increases in sympathetic drive (vasoconstriction, increased HR and myocardial contractility) can maintain arterial pressure within normal ranges under these circumstances. However, animals with mild to moderate degrees of hypovolemia may become severely hypotensive after induction of general anesthesia because drugs such as isoflurane decrease the central sympathetic outflow and blunt the baroreceptor reflex.
In dogs 20 mL/kg boluses of isotonic crystalloids (e.g., lactated Ringer's) administered over 15 minutes can be given to stabilize cardiovascular function during the peri-operative period. Artificial colloid solutions are more effective volume expanders than isotonic crystalloids and have the advantage of maintaining colloid oncotic pressure. In dogs, the volume of hetastarch necessary restore SAP to values observed before bleeding was close to the blood volume lost; contrasting with the volume of lactated Ringer's administered to achieve the same goal, that was 4 times greater than the blood volume lost (12). Because only 25% of an isotonic crystalloid remains within the intravascular compartment after 1 hour of its intravenous administration, the effective volume expansion achieved by a 20 mL/kg bolus lactated Ringer's is approximately 5 mL/kg. In hypovolemic dogs that are presented with perioperative hypotension 5 mL/kg boluses of pentastarch (Hemohaes®) or hetastarch may be administered over 15 minutes. Because cats have a smaller circulating volume (6% of body weight) than dogs (8% of body weight), crystalloids and artificial colloids should be given at slower rates in feline species.
Packed cell volume (PCV) and total plasma protein (TPP)/albumin are important ancillary tools for guiding volume replacement therapy in emergency situations. For dogs without heart disease presenting with PCV and TPP/albumin values within normal ranges prior to anesthesia, up to 2 isotonic crystalloid boluses (40 mL/kg) can be administered in addition to maintenance fluid therapy (10 mL/kg/hour of lactated Ringer's), without causing excessive hemodilution. Boluses of isotonic crystalloids should be avoided if plasma albumin is less than 2.0 g/dL (or TPP < 3.5 g/dL) because of increased risk of edema. In hypotensive animals that are hypoalbuminemic, artificial colloids (5 mL/kg boluses of pentastarch, up to a maximum of 20 mL/kg/day) may be used concomitantly with lower volumes of isotonic crystalloids. In animals receiving large volumes of fluids, acute decreases in PCV (< 20%) may compromise the blood's oxygen carrying capacity. Transfusion of packed red blood cells (or whole blood if the first option is not available) should be considered if PCV acutely falls below 20% in the presence of ongoing blood losses. Worsening of active bleeding during surgery may be caused by artificial colloids (dextrans, hetastarch) at volumes exceeding 20 mL/kg/day due to direct inhibition of platelet aggregation, or by large volumes of intravenous fluids used for volume replacement (dilutional coagulopathy); this condition may require the use of with fresh frozen plasma (10 mL/kg).
In most animals that are not severely ill, hypotension can be effectively treated by volume replacement therapy and by the use of a more balanced anesthetic technique. However in critically ill patients, hypovolemia is often associated with sepsis. Severe sepsis should be suspected if of hypotension persists after aggressive fluid therapy. Septic shock states are associated by low SVR and changes in microvascular permeability leading to blood flow maldistribution and hypotension.
Inotropes and vasopressors: Inotropes are drugs that primarily increase myocardial contractility while vasopressors are drugs that primarily increase SVR. These drugs are usually administered to animals that did not respond to volume replacement therapy. As increases in SVR may reduce the blood flow to splanchnic and peripheral organs, an increase arterial pressure should be preferably achieved by means of primarily increasing CO with the use of an inotrope. However there are circumstances (sepsis/systemic inflammatory response syndrome [SIRS]) where the use of a vasopressor may be desirable.
For relatively short anesthetic procedures in ASA I or II patients, if hypotension is not responsive 1-2 crystalloid boluses, ephedrine given as a bolus (0.1 to 0.2 mg/kg, IV) may be effective in restoring arterial blood pressure. Ephedrine increases CO by increasing myocardial contractility via beta-1 receptor stimulation and transiently increases SVR via alfa-1 receptor stimulation. Its duration of action after a single bolus administration is of approximately 15-60 minutes.8,13
Dopamine is commonly used to stabilize blood pressure in small animal patients undergoing major surgery. Dopamine is a catecholamine with dose-related hemodynamic effects: at medium doses (5-10 mcg/kg/min) dopamine acts mainly as an inotrope; at higher doses (> 10-15 mcg/kg/min) dopamine acts as an inotrope and as a vasopressor. In isoflurane-anesthetized dogs, dopamine increased CO (beta-1 receptor stimulation) and caused a biphasic change in SVR: at doses of 5-10 mcg/kg/min SVR was decreased, later returning to baseline at doses of 15 mcg/kg/min.8 An increase in MAP was only evident at the highest dose. Clinical experience has shown that dopamine at doses ranging from 5 to 15 mcg/kg/min are necessary normalize arterial pressure in hypotensive patients. Higher doses may be necessary if hypotension is associated with excessive vasodilation. Dopamine is the author's first choice in most critically ill patients (ASA III to V) that do not respond to volume replacement therapy because the infusion rate can be easily titrated to effect.
Critically ill patients with hypotension due to decreased cardiac contractility (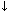 CO) and vasodilation (
CO) and vasodilation (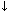 SVR) may benefit of the effects of dopamine at higher doses (
SVR) may benefit of the effects of dopamine at higher doses (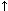 CO and
CO and 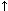 SVR). However, a more potent vasopressor may be necessary in patients with hypotension due to sepsis/systemic inflammatory response syndrome. Norepinephrine has greater affinity for alfa-1 receptors than dopamine; doses ranging from 0.1 to 0.3 mcg/kg/min are effective in improving renal blood flow and in restoring arterial blood pressure in hypotensive dogs with severe sepsis.14 Because norepinephrine induced vasoconstriction (
SVR). However, a more potent vasopressor may be necessary in patients with hypotension due to sepsis/systemic inflammatory response syndrome. Norepinephrine has greater affinity for alfa-1 receptors than dopamine; doses ranging from 0.1 to 0.3 mcg/kg/min are effective in improving renal blood flow and in restoring arterial blood pressure in hypotensive dogs with severe sepsis.14 Because norepinephrine induced vasoconstriction (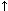 SVR) has the potential for restricting peripheral and splanchnic blood flow, this vasopressor should be carefully titrated to achieve a SAP between 90 to 100 mm Hg (or a MAP of 60-70 mm Hg) and its use should be restricted to clinical cases where other attempts to treat hypotension have failed. Norepinephrine infusion rate can be started at 0.1 mcg/kg/min; further adjustments in the infusion rate are performed to maintain arterial blood pressure within the desired range.
SVR) has the potential for restricting peripheral and splanchnic blood flow, this vasopressor should be carefully titrated to achieve a SAP between 90 to 100 mm Hg (or a MAP of 60-70 mm Hg) and its use should be restricted to clinical cases where other attempts to treat hypotension have failed. Norepinephrine infusion rate can be started at 0.1 mcg/kg/min; further adjustments in the infusion rate are performed to maintain arterial blood pressure within the desired range.
In patients with end-stage hypovolemic/hemorrhagic shock, sepsis, and SIRS, hypotension may persist in spite of volume replacement therapy and of the use of catecholamines. Vasopressin (1-4 mcg/kg/min) was shown to restore arterial pressure to normal values in cases where catecholamines were no longer effective.15 Vasopressin should not be used as the sole vasopressor and prognosis for long term survival is poor if vasopressin is the only effective drug for treating hypotension in critically ill patients.
References
1. Beck JJ, Staatz AJ, Pelsue DH, et al. J Am Vet Med Assoc. 2006;229:1934-1939.
2. Silverstein DC, Wininger FA, Shofer FS, et al. J Am Vet Med Assoc 2008;232:893-897.
3. Wagner AE, Hellyer PW. J Vet Med Educ 2002;29:176-182.
4. Deflandre CJA, Hellebrekers LJ, et al. Vet Anaesth Analg 2008;35-13-21.
5. Dyson D. J Vet Emerg Crit Care 2007;17:135-142.
6. Caulkett NA, Cantwell SL, Houston DM. Vet Surg 1998;27:370-7.
7. Monteiro ER, Teixeira Neto FJ, Castro VB, et al. Vet Anaesth Analg 2007;34:312-321.
8. Sinclair M, Dyson DH. Vet Anaesth Analg 2009;36:1 (Abstract).
9. Ilkiw JE, Pascoe PJ, Haskins SC, et al. Can J Vet Res 1994;58:248-253
10. Steagall PVM, Teixeira Neto FJ, Minto BW, et al. J Am Vet Med Assoc 2006;229:522-527
11. Dyson DH, James-Davies R. Can Vet J 1999;40:327-331.
12. Muir WW, Wiese AJ. Am J Vet Res 2004;65:1189-1194.
13. Wagner AE, Dunlop CI, Chapman PL. Am J Vet Res 1993;54:1917-1922.
14. Bellomo R, Kellum JA, Wisniewski SR, et al. Am J Respir Crit Care Med 1999;159:1186-1192.
15. Morales D, Madigan J, Cullinane S, et al. Circulation. 1999;100:226-229.