Theresa W. Fossum, DVM, PhD, DACVS
Tom and Joan Read Chair in Veterinary Surgery, Director, Clinical Programs and Biomedical Devices, Michael E. DeBakey Institute Professor of Surgery, Texas A&M University College of Veterinary Medicine
College Station, TX, USA
Diaphragmatic hernias are commonly recognized by small animal clinicians and may be congenital or occur secondary to trauma. Congenital pleuroperitoneal hernias are seldom diagnosed in small animals because many affected animals die at birth or shortly thereafter. Most diaphragmatic hernias in dogs and cats are caused by trauma, particularly motor vehicle accidents. The abrupt increase in intraabdominal pressure accompanying forceful blows to the abdominal wall causes the lungs to rapidly deflate (if the glottis is open), producing a large pleuroperitoneal pressure gradient. Alternately, the pressure gradient that occurs between the thorax and the abdomen may cause the diaphragm to tear. The tears occur at the weakest points of the diaphragm, generally the muscular portions. Traumatic diaphragmatic hernias are often associated with significant respiratory embarrassment; however, chronic diaphragmatic hernias in asymptomatic animals are not uncommon.
Traumatic Diaphragmatic Hernias
Clinical Presentation
Signalment
There is no breed predisposition for traumatic diaphragmatic hernias. Young males have historically thought to be more commonly affected; however, a recent study of traumatic diaphragmatic hernias identified no sex predilection.
History
The duration of a diaphragmatic hernia may range from a few hours to years. Many (15% to 25%) are diagnosed weeks after the injury. The animals may be presented in shock acutely after the trauma, or the hernia may be an incidental finding. Animals sustaining trauma often suffer from associated injuries (e.g., fractures). With a chronic diaphragmatic hernia, the clinical signs most often are referable to either the respiratory (i.e., dyspnea, exercise intolerance) or the gastrointestinal systems (i.e., anorexia, vomiting, diarrhea, weight loss, pain after ingestion of food) or they may be nonspecific (e.g., depression). Many animals with chronic hernias are not dyspneic at the time of diagnosis.
Physical Examination Findings
Animals with recent traumatic diaphragmatic hernias frequently are in shock when presented for treatment; therefore, clinical signs may include pale or cyanotic mucous membranes, tachypnea, tachycardia, and/or oliguria. Cardiac arrhythmias are common and associated with significant morbidity. Other clinical signs depend on which organs have herniated and may be attributed to the gastrointestinal, respiratory, or cardiovascular system. The liver is the most commonly herniated organ, a condition that often is associated with hydrothorax caused by entrapment and venous occlusion.
Diagnostic Imaging
Definitive diagnosis of pleuroperitoneal diaphragmatic hernia usually is made by radiography or ultrasonography. It may be difficult to diagnose diaphragmatic hernias radiographically if only a small portion of the liver is herniated. In a recent study, thoracic radiographs revealed evidence of diaphragmatic hernia in only 66% of affected animals. Ultrasound examination of the diaphragmatic silhouette may help when herniation is not obvious radiographically (i.e., hepatic herniation, pleural effusion).
Positive contrast celiography occasionally may be helpful. Prewarmed water-soluble iodinated contrast agent is injected into the peritoneal cavity at a dosage of 1.1 ml/kg (the dose is doubled if ascites is present), the patient is gently rolled from side to side or the pelvis is elevated, and films are taken immediately after the injection and manipulation. Criteria used in evaluating these images should include the presence of contrast medium in the pleural cavity, absence of a normal liver lobe outline in the abdomen, and incomplete visualization of the abdominal surface of the diaphragm. Positive-contrast celiograms should be interpreted cautiously, because omental and fibrous adhesions may seal the defect, resulting in false negative studies.
Medical Management
If the animal is dyspneic, oxygen should be provided by face mask, nasal insufflation, or an oxygen cage. Positioning the animal in sternal recumbency with the forelimbs elevated may help ventilation. If moderate or severe pleural effusion is present, thoracentesis should be performed. Fluid therapy and antibiotics should be given if the animal is in shock.
Surgical Treatment
Chronic diaphragmatic hernias may have a higher mortality than acute diaphragmatic hernias; however, the prognosis with both groups is good to excellent with surgery. If pulmonary contusions are severe, surgical repair of diaphragmatic hernias should be delayed until the patient's condition has been stabilized; however, herniorrhaphy should not be delayed unnecessarily. Animals with gastric herniation should be evaluated carefully for gastric distention and should be operated on as soon as they can safely be anesthetized, because acute gastric distention within the thorax may cause rapid, fatal respiratory impairment.
Preoperative Management
Prophylactic antibiotics should be given before induction of anesthesia in animals with hepatic herniation. Massive release of toxins into the circulation may occur with hepatic strangulation or vascular compromise. Premedicating such patients with steroids may be beneficial. An ECG should be performed on all trauma patients before surgery.
Surgical Technique
Make a ventral midline abdominal incision; if greater exposure is needed, extend the incision cranially through the sternum. Replace the abdominal organs in the abdominal cavity (if necessary, enlarge the diaphragmatic defect). If adhesions are present, dissect the tissues gently from the thoracic structures to prevent pneumothorax or bleeding. With chronic hernias, debride the edge of the defect before closure. Close the diaphragmatic defect in a simple continuous suture pattern. If the diaphragm is avulsed from the ribs, incorporate a rib in the continuous suture for added strength (Figure 1). Remove air from the pleural cavity after closing the defect. If continued pneumothorax or effusion is likely, place a chest tube. Explore the entire abdominal cavity for associated injury (i.e., compromise of the vasculature to the intestine or splenic, renal, or bladder trauma) and repair any defects.
| Figure 1. | 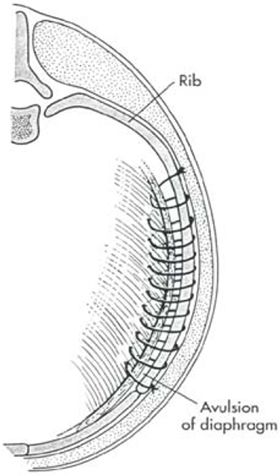
From: Fossum, TW: Small Animal Surgery, Mosby Publishing Co., St. Louis, Mo, 2002 |
|
| |
Peritoneopericardial Diaphragmatic Hernia
Peritoneopericardial diaphragmatic hernias are less commonly recognized by small animal clinicians than traumatic diaphragmatic hernias. Although PPDH often are associated with respiratory embarrassment, asymptomatic PPDH is common. PPDH may occur as a result of trauma in human beings (in whom the diaphragm forms one wall of the pericardial sac); however, these hernias are always congenital in dogs and cats, in which no direct communication exists between the pericardial and peritoneal cavities after birth. The most widely accepted theory regarding the embryogenesis of this defect is that the hernia occurs because of faulty development or prenatal injury of the septum transversum. This could be a result of a teratogen, genetic defect, or prenatal injury.
Clinical Presentation
Signalment
Although PPDH is congenital, it is not uncommon for the diagnosis to be made when the animal is middle-aged or older because clinical signs vary and may be intermittent. Weimaraners and cocker spaniels may be at increased risk. Domestic longhair and Himalayan cats may be predisposed.
History
The clinical signs may be referable to the gastrointestinal, cardiac, or respiratory systems and include anorexia, depression, vomiting, diarrhea, weight loss, wheezing, dyspnea, exercise intolerance, and/or pain after eating. Neurologic signs may occur as a result of hepatoencephalopathy.
Surgical Technique
Make a ventral midline abdominal incision. If greater exposure is needed, extend the incision cranially through the sternum. Enlarge the diaphragmatic defect if necessary and replace the abdominal organs in the abdominal cavity. If adhesions are present, gently dissect the tissues from the thoracic structures, resecting or debriding necrotic tissue as necessary. Debride the edges of the defect and close in a simple continuous suture pattern. Do not close the pericardial sac. Remove air from the pericardial sac or pleural cavity or both after closing the defect. If continued pneumothorax or effusion is likely, place a chest tube. Repair concomitant sternal or abdominal wall defects.
Prognosis
If the animal survives the early postoperative period (i.e., 12 to 24 hours), the prognosis is excellent, and recurrence is uncommon with proper technique. A postoperative mortality rate of 14% was recently reported in cats. The prognosis is worse in patients with PPDH that have concurrent cardiac abnormalities.