Development of a Canine Trypsin-like Immunoreactivity Assay System Using Monoclonal Antibodies
Exocrine pancreatic insufficiency (EPI) often occurs in dogs that are obese and/or have hereditary predisposing factors. Severe atrophy of the exocrine pancreas is the most common cause and leads to the typical gross pathologic findings of thin and transparent tissue with visible abnormal glandular structures. Several indirect pancreatic function tests are used to diagnose end-stage EPI.
To diagnose EPI in veterinary clinics, the detection of undigested food in feces, assay of fecal proteolytic enzyme activity, starch tolerance test, and assessment of fat absorption are the most usual methods. Unfortunately, these tests frequently are misleading or unhelpful because many of the results are equivocal. Although assaying the pancreatic enzyme activity in intestinal fluid after oral administration of the chymotrypsin substrate bentiromide (N-benzoyl-L-p-aminobenzoic acid; BT-PABA) is a reliable method, the test is time-consuming and tasks the patient.
Because trypsinogen is synthesized and stored only in pancreatic acinar cells, serum trypsin-like immunoreactivity (TLI), which reflects the concentration of both trypsinogen and trypsin, is a pancreas-specific enzyme and a marker for exocrine pancreatic function. The radioimmunoassay (RIA) for TLI is one of the most sensitive and specific tests for diagnosing EPI in dogs. Healthy dogs have a serum TLI concentration of 5.0 to 35.0 ng/ml, whereas dogs with end-stage EPI have abnormally low (< 2.5 ng/ml) levels (Williams and Batt, 1983, 1986, 1988). Prior to the present study, we could detect canine serum TLI values only by RIA, but these tests are beyond the capabilities of most veterinary clinics and general laboratories. Using monoclonal antibodies (mAbs) that recognize canine TLI, we developed an ELISA method for TLI and incorporated it into an immuno chromatographic test (ICT) for the diagnosis of canine EPI.
Materials and methods
ELISA for canine TLI
Canine cationic trypsin was purified as described previously (Waritani et al., 2001). Purified cationic trypsin was irreversibly inactivated with diisopropyl fluorophosphate (DFP) which is protease inhibitor. The mAb against canine TLI (reagent 009-303) was prepared as previously described (Waritani et al., 2001). The F(ab')2 and Fab fractions of 009-303 were obtained by pepsin digestion followed by 2-aminoethanethiol reduction. The Fab fraction of 009-303 was conjugated with peroxidase (POD) by using N-(6-maleimidocaproyloxy)-succinimide.
A 1-step sandwich ELISA method was developed using mAb against canine TLI (reagent 008-207) (Waritani et al., 2001) and the POD-conjugated Fab of 009-303. 008-207 (100 µg/ml in 0.1 M Na-phosphate buffer [pH 7.5]; 100 µl per well) was used to coat the wells of a 96-well microplate. PBS (pH 7.0) containing 0.05% Tween-20 (TPBS), 0.1M Na-phosphate buffer and POD-conjugated Fab of 009-303 MAb were used for washing, for blocking and for second antibody respectively. For standards, 5 µl DFP-inactivated cationic trypsin (1 to 100 ng/ml in PBS-BSA) was added to each well; patient sample wells received 5 µl canine serum. A 2,2'-azinobis (3-ethylbenzthiazoline) sulfonic acid was used as a substrate following the manufacturer's instructions. The absorbance in the wells was measured by using a microplate reader with filter at 405 nm / 492 nm (the side of sample / reference) .
Preparation of an ICT kit for canine TLI
The ICT was developed by modifying a previously described technique (Snowden and Hommel, 1991; De Mey et al., 1981). Briefly, 0.05 to 1 µg 008-207 and 2 µg anti-mouse IgG polyclonal antibody were bound to a 8 × 25-mm nitrocellulose strip to generate sample and control lines, respectively. After blocking with 0.01 M carbonate-phosphate buffer containing 1% BSA, the strips were dried at room temperature. Next, 100 µg 009-303 was mixed with 1 ml of gold colloid solution ( British bio cell international, UK) (pH 9.0) at room temperature. After 30 min, BSA was added to 1% of the final concentration. The mixture was centrifuged at 8000 × g for 30 min at 4 °C. The precipitated gold colloid-conjugated 009-303 was dissolved in 0.01 M Tris-buffered saline containing 1% BSA; this dissolved solution was absorbed into the conjugate pad, which was then dried at 58 °C. The ICT kit thus comprised a nitrocellulose strip, a conjugate pad, and an absorbent pad in a plastic case.
To perform the assay, 1 drop of canine serum and 3 drops of reaction buffer (0.05 M PBS [pH 8.0] containing 0.05% Tween-20) are added to the sample well of the case. The TLI in canine serum flows through the absorbent pad and reacts with the gold colloid-conjugated 009-303 in the conjugate pad. The immunocomplex of TLI-gold colloid-conjugated 009-303 moves by capillary action to the nitrocellulose strip. The immunocomplex reacts to the 008-207 on the sample line and is immobilized there. The excess gold colloid-conjugated 009-303 reacts to the anti-mouse IgG polyclonal antibody on the control line, and the resulting complex is immobilized there. If the procedure has been carried out correctly, a red control line appears after 10 min. If a red sample line also appears (i.e., a positive result), the TLI value of the sample is >5 ng/ml, and the dog does not have EPI. If a control line but no sample line appears (i.e., a negative result), the TLI value of the sample is <2 ng/ml, and the dog most likely has EPI. The canine TLI ICT kit is produced and supplied commercially by Daiichi Fine Chemical Co., Ltd. (Takaoka, Japan).
Normal canine serum samples were obtained from 65 clinically healthy dogs. Test samples (n = 41) were collected from dogs that had been diagnosed with pancreatitis (n = 12), EPI (n = 6), or gastrointestinal disease (n = 23) by history, physical examination, and biochemical profile and various combinations of ancillary tests including abdominal radiographs, ultrasonography, and histopathology of pancreas tissue.
Serum TLI values by RIA were determined by using a double-antibody canine TLI RIA kit according to the manufacturer's instructions (DPC; Diagnostic products corporation, Los Angeles, CA, USA). Statistical analyses of the data were performed by using Student's t test. Correlation coefficient was calculated by Microsoft Excel.
Results
The sensitivity of the ELISA method was 0.5 ng/ml TLI. Linearity was observed over 1 to 100 ng/ml TLI for the combination of 008-207 as the solid phase and the POD-conjugated Fab of 009-303 as the secondary antibody. Serum samples from clinically healthy dogs were used to determine the levels of intra- and inter-assay coefficients of variance (CVs). The intra-assay CVs ranged from 1.8% to 6.1% (n = 5) in samples with TLI concentrations of 6.9 to 23.9 ng/ml. Inter-assay CVs were 5.1% to 9.8% (n = 5) (Table 2). The recovery of TLI added to 2 samples of serum ranged from 89% to 111% and from 93% to 108% (n = 5) in two samples.
The TLI values obtained by ELISA for samples from 56 dogs (healthy dogs 20, patients dog 36) correlated well (correlation coefficient, 0.974) with those obtained by RIA. Serum samples collected from 3 healthy dogs were diluted serially twofold using fetal calf serum. The TLI values of the diluted sera were evaluated by both ELISA and ICT kit. The ICT kit yielded positive (i.e., no EPI) results for TLI concentrations exceeding 5.3 ng/ml and negative results (i.e., EPI suspected) when the TLI level was lower than 1.8 ng/ml. Because the cut-off value of the ICT kit was between 2.0 and 5.0 ng/ml TLI, the kit returned variable results for samples with 3.1 to 3.3 ng/ml TLI. For the all canine samples (n=106) tested, the ICT kit returned negative results when TLI were <3.4 ng/ml by ELISA and positive results when the ELISA-determined TLI concentration was >4.1 ng/ml. The serum TLI concentrations of the samples returning positive results differed significantly (P < 0.01) from those of samples yielding negative results (Fig.1).
By ELISA, the serum TLI concentration (mean ± 1 standard deviation) of the 65 clinically healthy dogs was 17.0 ± 5.3 (range, 7.8 to 29.2) ng/ml. For the 12 dogs diagnosed with pancreatitis, the average serum TLI value was 63.9 ± 89.8 (range, 0.0 to 287) ng/ml, and that for the 23 dogs with gastroenteritis was 18.9 ± 18.3 (range, 3.0 to 71.8) ng/ml. However, the TLI concentration was 0.3 ± 0.2 (range, 0 to 0.7) ng/ml in the 6 dogs with EPI (Fig. 5). The TLI concentrations of the clinically healthy dogs differed significantly (P < 0.01) from those of the animals with EPI. But TLI values did not differ between clinically healthy dogs and those with pancreatitis (P = 0.134) or gastroenteritis (P = 0.825). A positive ICT sample line did not occur when the TLI concentration of the canine serum sample was < 4.1 ng/ml by ELISA (Fig.2). Further, all 6 dogs diagnosed with EPI were negative (no sample line) by ICT, as were 3 of the 12 dogs with pancreatitis and 1 of the 23 dogs with gastrointestinal disease.
DISCUSSION
The present study indicates that our ELISA method and ICT have high sensitivity and specificity for the diagnosis of canine EPI. These system do not use radio isotope, then they can be used everywhere and every time for assist of veterinary clinics. Now, we are also trying to develop an ICT for the diagnosis of canine pancreatitis.
| 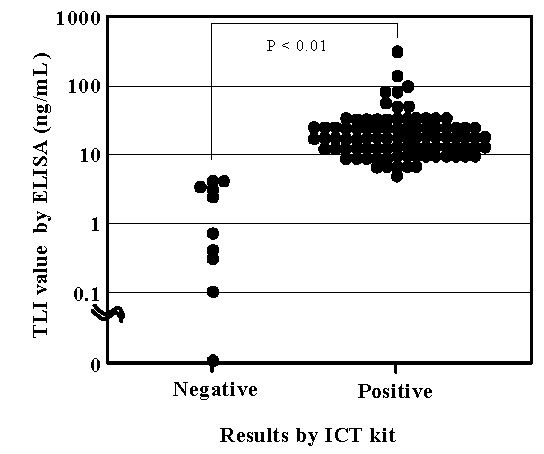
Figure 1. Correlation between results of ICT kit and ELISA-determined serum TLI concentration. |
|
| |
| 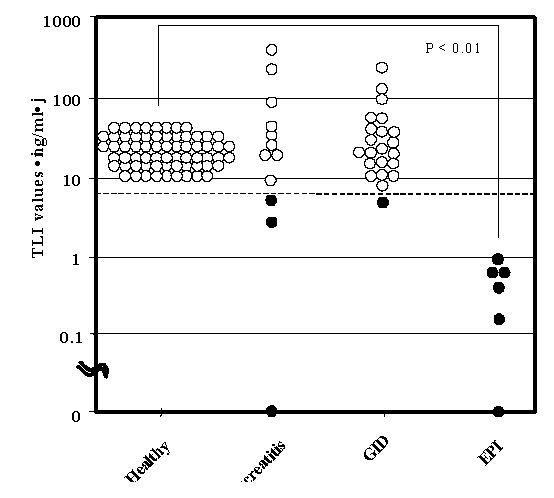
Figure 2. ELISA-determined serum TLI values for healthy and disease dogs. The dashed line indicates a serum TLI of 5ng/ml, the cut-off value for the diagnosis of EPI. Positive(? ) and negative(? ) results from ICT are from the same samples. |
|
| |