Abstract
Mycobacterium orygis, a member of the Mycobacterium tuberculosis complex (MTBC), has been isolated predominantly from hoofstock in eastern Africa and the Arabian Peninsula, and occasionally in cattle, rhesus monkeys, humans, and greater one-horned (GOH) rhinoceros (Rhinoceros unicornis) in southern Asia, with documented zoonotic and zooanthroponotic transmission.1,5,7 Typically, rhinoceros develop chronic progressive respiratory disease as a result of MTBC infection.3 This report describes the postmortem diagnosis of tuberculosis caused by M. orygis in a GOH rhinoceros imported from India who developed hindlimb paresis due to neural granulomatosis. The postmortem use of dual path platform technology (DPP® VetTB Assay)a and multi-antigen print immunoassays (MAPIA)a to detect specific anti-MTBC antibodies prior to culture results on serum collected just before death from this rhinoceros allowed for appropriate preventive and management protocols to be initiated in the herd, highlighting the potential of serodiagnostics for antemortem diagnosis of tuberculosis in rhinoceros (Figures 1 and 2).2,8 Mycobacterium genus-specific PCR assaysb followed by direct sequencing allowed timely confirmation of the serology results.4,6 Banked serum collected up to 6 yr prior to death correlated closely with the course of clinical disease, suggesting latent infection originating from India.9 This is the first isolation of M. orygis within the western hemisphere, further exemplifying the need for more stringent testing prior to international shipment, and the urgency for validated antemortem MTBC screening assays in rhinoceros species.
Endnotes
a. Chembio Diagnostic Systems, Inc, 3661 Horseblock Road, Medford, NY, USA
b. Washington Animal Disease Diagnostic Lab (WADDL), College of Veterinary Medicine, Washington State University, Bustad Hall Room 155N, Pullman, WA, USA
Acknowledgments
The authors would like to thank veterinary and animal management staff at The Wilds for their incredible care of this rhinoceros prior to its death. The authors also acknowledge the staff and laboratory technicians at Washington Animal Disease Diagnostics Laboratory and National Veterinary Services Laboratory for their diligent work with the sample testing for this case.
Figure 1

Antibody response against eight recombinant Mycobacterium tuberculosis complex (MTBC) protein antigens (ESAT6, CFP10, MPB64, MPB70, MPB83, E6/P10, DID38, and DID65) and two native antigens (M. bovis purified protein derivative, B-PPD, and M. bovis culture filtrate, MBCF) detected by multi-antigen print immunoassay (MAPIA) in serum samples collected between 2010 and 2016 from a greater one-horned rhinoceros bull diagnosed in 2016 with tuberculosis from M. orygis infection.
Figure 2
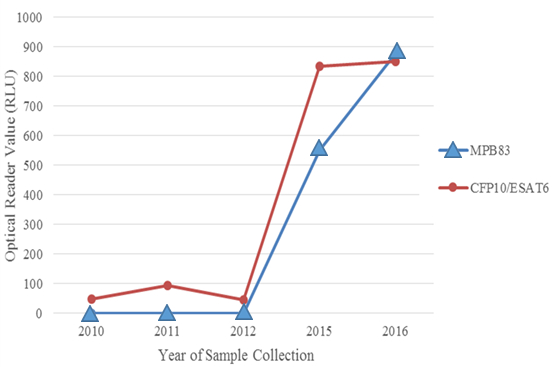
Evolution of antibody response against the major Mycobacterium tuberculosis complex protein antigens, MPB83 and CFP10/ESAT6, detected by DPP VetTB assay in serum samples collected between 2010 and 2016 from a greater one-horned rhinoceros diagnosed in 2016 with tuberculosis from M. orygis infection. Optical reader value (Y-axis) reflectance measured in relative light units (RLU); broken line indicates cut-off value of 40 RLU, equal to threshold of visual test interpretation.
Literature Cited
1. Dawson KL, Bell A, Kawakami RP, Coley K, Yates G, Collins DM. Transmission of Mycobacterium orygis (M. tuberculosis complex species) from a tuberculosis patient to a dairy cow in New Zealand. J Clin Microbiol. 2012;50:3136–3138.
2. Miller MA, Greenwald R, Lyashchenko KP. Potential for serodiagnosis of tuberculosis in black rhinoceros (Diceros bicornis). J Zoo Wildl Med. 2015;46:100–104.
3. Miller M, Michel A, Helden P van, Buss P. Tuberculosis in rhinoceros: an underrecognized threat? Transbound Emerg Dis. 2016.
4. Roth A, Fischer M, Hamid ME, Michalke S, Ludwig W, Mauch H. Differentiation of phylogenetically related slowly growing mycobacteria based on 16S–23S rRNA gene internal transcribed spacer sequences. J Clin Microbiol. 1998;36:139–147.
5. Thapa J, Paudel S, Sadaula A, Shah Y, Maharjan B, Kaufman GE, McCauley D, Gairhe KP, Tsubota T, Suzuki Y, Nakajima C. Mycobacterium orygis-associated tuberculosis in free-ranging rhinoceros, Nepal, 2015. Emerg Infect Dis. 2016;22:570–572.
6. van Ingen J, de Zwaan R, Dekhuijzen R, Boeree M, van Soolingen D. Region of difference 1 in nontuberculous Mycobacterium species adds a phylogenetic and taxonomical character. J Bacteriol. 2009;191:5865–5867.
7. van Injen J, Rahim Z, Mulder A, Boeree MJ, Simeone R, Brosch R, van Soolingen D. Characterization of Mycobacterium orygis as M. tuberculosis complex subspecies. Emerg Infect Dis. 2012;18:653–655.
8. Wadhwa A, Hickling GJ, Eda S. Opportunities for improved serodiagnosis of human tuberculosis, bovine tuberculosis, and paratuberculosis. Vet Med Int. 2012;2012:1–13.
9. Waters WR, Palmer MV, Bannantine JP, Whipple DL, Greenwald R, Esfandiari J, Andersen P, McNair J, Pollock JM, Lyashchenko KP. Antigen recognition by serum antibodies in white-tailed deer (Odocoileus virginianus) experimentally infected with Mycobacterium bovis. Clin Vaccine Immunol. 2004;11(5):849–855.