Liver Enzymes vs. Liver Function Tests
Liver test abnormalities can be categorised loosely into markers of hepatocellular injury, cholestasis, or impaired liver function.
Transaminases ALT and AST
ALT is a cytoplasmic enzyme of hepatocytes that is more specific than AST. ALT has a half-life of 6 hours in a cat and 48–60 hours in a dog, making it a good indicator of continued liver injury.1 AST is a mitochondrial-bound enzyme found in high concentrations in hepatocytes. Consequently, its increase indicates cell death. However, AST is also found in myocytes and red blood cells. The half-life is 77 minutes in a cat and 22 hours in a dog.1
Indicators of Cholestasis
ALKP and GGT typically increase with cholestatic disease or drug induction. Cholestasis increases the surface tension of the bile canaliculi and ductules, inducing enzyme production. ALKP has a half-life of 66–70 hours in dogs and 6 hours in cats.1 Apart from being hepatobiliary in origin, increased bone isoenzyme may occur in young animals or those with lytic or neoplastic bone lesions. In dogs, ALKP increase is also seen with endogenous or exogenous corticosteroids and anti-epileptic drugs (AEDs), whereas GGT does not increase with bone activity and AEDs.
Liver Function
Liver failure can manifest as decreased synthetic capacity, decreased uptake and excretion of bile acids, and decreased conversion of ammonia to urea. For example, laboratory evidence of decreased synthetic capacity can occur when 70–75% of the hepatic functional mass is lost. Hepatic dysfunction may manifest as decreased albumin, glucose, cholesterol and BUN. Production of coagulation factors, fibrinogen, as well as coagulation inhibitors (e.g., protein C and anti-thrombin) are also affected.
Bile acids are synthesised from cholesterol in the liver and secreted in bile after conjugation. Assessing the liver’s capacity to remove circulating bile acids gives an indication of liver function and perfusion. Its use in detection of hepatobiliary disease and acute liver damage in dogs, as well as portosystemic vascular anomalies (PSVA) in dogs and cats, has been described across various studies.2-4 Understanding the limitations of an endogenous challenge helps in the interpretation of unusual test results (e.g., higher preprandial concentrations). One should also be aware of test vulnerability factors including meal type and size, enteric disease and intestinal motility.5 Lastly, one should also bear in mind the possible effects of concurrent administration of ursodeoxycholic acid, as well as the spurious increase in bile acids seen in Maltese terriers.6,7 Fasting ammonia or the ammonia tolerance test serves as possible alternatives to detect hepatic dysfunction and PSVAs. It is less commonly used due to complexities in sample handling.
Protein C is a vitamin K-dependent protein, synthesised in the liver with a short plasma half-life. It serves as an indicator of portal blood flow. It may be used to differentiate PSVA from microvascular dysplasia (MVD), as both disorders may result in increased bile acids.8 Low protein C activity (<70%) is a characteristic finding in dogs with clinical signs of PSVA. In contrast, dogs with MVD typically have protein C values >70%. Protein C deficiency, however, may also be seen in patients with vitamin K deficiency, sepsis, SIRS, and DIC.9
Approach
| Figure 1. Approach to increased liver enzymes10 | 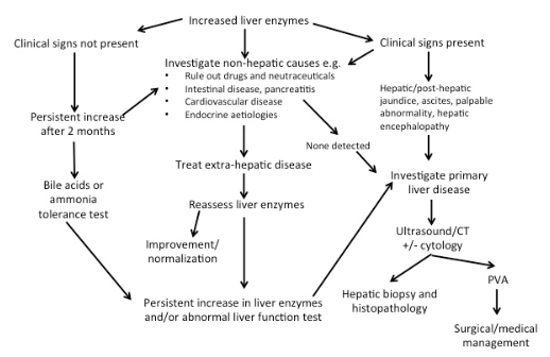
|
|
| |
The decision to investigate is difficult in asymptomatic animals. If no drug history (including nutraceuticals) is found, persistent increase of 2 months’ duration or progression within this period is generally an indication for further testing. The speed at which investigation takes place may further be influenced by the patient’s signalment, in conjunction with known breed predispositions.
Investigation is recommended when clinical signs are present, especially if the signs are specific to hepatic disease. Again, history, physical examination and signalment are all critical in establishing the cause of the increased enzymes. Based on clinical signs, extrahepatic disease should be investigated if indicated. These include intestinal, respiratory and cardiovascular diseases, which generally result in mild to moderate increase in transaminase activity if present. Endocrinopathies (e.g., hyperthyroidism in cats), hypothyroidism and hyperadrenocorticism in dogs should be considered in the presence of appropriate clinicopathological abnormalities. Investigation of primary hepatic disease is indicated if hepatic enzymes remain persistently increased despite addressing extrahepatic disorders.
Investigation of primary liver disease may involve assessment of hepatic function, imaging and +/- histopathology. Ultrasound is a commonly used modality to image the liver, although scintigraphy and computed tomography are generally thought to be more sensitive for detection of PSVAs. However, histopathology is often required for definitive diagnosis, prognosis and to dictate ongoing management.
Cases
References
1. Cocker S, Richter K. Hepatobiliary disease. In: Ettinger SJ, Feldman EC, eds. Textbook of Veterinary Internal Medicine: Diseases of the Dog and Cat. Philadelphia: WB Saunders Co; 2017:1611–1621.
2. Center SA, Manwarren T, Slater MR, Wilentz E. Evaluation of twelve-hour preprandial and two-hour postprandial serum bile acids concentrations for diagnosis of hepatobiliary disease in dogs. J Am Vet Med Assoc. 1991;199:217–226.
3. Turgut K, Demir C, Ok M, Ciftçi K. Pre- and postprandial total serum bile acid concentration following acute liver damage in dogs. Zentralbl Veterinarmed A. 1997;44:25–29.
4. Dirksen K, Burgener IA, Rothuizen J, van den Ingh TSGAM, Penning LC, Spee B, Fieten H. Sensitivity and specificity of plasma ALT, ALP, and bile acids for hepatitis in Labrador retrievers. J Vet Intern Med. 2017;31:1017–1027. doi: 10.1111/ jvim.14716.
5. Center SA, Randolph JF, Warner KL. Influence of oral ursodeoxycholic acid on serum and urine bile acid concentrations in clinically normal dogs. ACVIM 2004.
6. Tisdall PL, Hunt GB, Tsoukalas G, Malik R. Post-prandial serum bile acid concentrations and ammonia tolerance in Maltese dogs with and without hepatic vascular anomalies. Aust Vet J. 1995;72:121–126.
7. Abraham LA, Charles JA, Holloway SA. Effect of oral ursodeoxycholic acid on bile acids tolerance tests in healthy dogs. Aust Vet J. 2004;82:157–160.
8. Toulza O, Center SA, Brooks MB, Erb HN, Warner KL, Deal W. Evaluation of plasma protein C activity for detection of hepatobiliary disease and portosystemic shunting in dogs. J Am Vet Med Assoc. 2006;229:1761–1771.
9. de Laforcade AM, Rozanski EA, Freeman LM, Li W. Serial evaluation of protein C and antithrombin in dogs with sepsis. J Vet Intern Med. 2008;22:26–30.
10. Twedt DC. Abnormal liver enzymes: a practical clinical approach. Colorado State University; [Last accessed 05 June 2018]. Canine/feline liver & pancreatic disease. Available from: https://pdfs.semanticscholar.org/8319/b718031aa9dedaf57e23d719d8e90353b799.pdf.