Abstract
The eyes of teleost fish follow the basic vertebrate blueprint consisting of scleral, uveal, and neural layers, a crystalline lens, and clear media (aqueous and vitreous humors).6 In the least derived species, nutrition to the posterior segment is provided solely via diffusion from the choroid. Most teleosts possess an outgrowth of the choroid into the vitreous humor, termed the falciform process, as well as a vascular rete mirabile (originally and mistakenly termed the “choroidal gland”), which receives oxygenated blood from the pseudobranch and is thought to enhance oxygen tension in the posterior segment.1,3,7,8 The choroid of the black pacu or tambaqui (Colossoma macropomum), a relative of the piranha and native to the Amazon River basin, also contains a thick pad of adipose tissue (C. Murphy, unpublished data). This presence of fat in the choroid appears to be nearly unique among vertebrates, as yet undocumented in the published literature, and of unknown function. This study correlates findings from imaging of intact pacu with gross and histologic anatomic features.
Seven black pacu averaging approximately 13 kg were examined in this study. Three fish were anesthetized using tricaine methane sulfonate (MS-222) for ultrasonographica imaging of the eyes. Four additional fish were euthanized using an overdose of MS-222. Computed tomographicb images were made immediately post-mortem on three fish, and high-resolution magnetic resonancec images were made of exenterated eyes of one fish. In all modalities, the cornea, anterior chamber, lens, vitreous humor, and retina were identified and had an appearance typical of the vertebrate eye, with the exception that the retina was located at approximately half of the anterior-posterior (A-P) depth of the globe and had a flatter contour than the posterior sclera (Figure 1). The optic nerve was visualized well in all modalities except ultrasound. In all modalities, the majority of the globe posterior to the retina had an appearance consistent with fat. The choroid rete was identified within this fatty region in all specimens; as is typical of many teleosts,6 it took the shape of an inverted U dorsal to the optic nerve head.
Figure 1. Ocular anatomy of the black pacu (Colossoma macropomum)
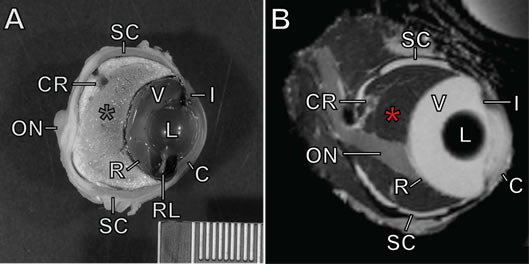
A) Formalin-fixed eye in parasagittal section. B) MRI image of eye. See text for methods. C=cornea; I=iris; L=lens; V=vitreous humor; RL=retractor lentis muscle; SC=scleral cartilages; R=retina; CR=choroid rete mirabile (dorsal portion seen cross section); ON=optic nerve. Red asterisk=choroid, occupying the space posterior to the retina and anterior to the sclera. Scale bar=1 mm hatches.
The eyes of five of the above animals were fixed in formalin for gross and histologic examination. Eyes were decalcified and sectioned parasagittally, photographed, and embedded in paraffin. Sections were stained with hematoxylin and eosin. Slides from two archived specimens (courtesy D. Dubielzig) were also examined. The cornea, iris, lens, retractor lentis muscle, falciform process, retina, optic nerve, choroidal rete, and sclera (including scleral cartilage and ossicles) were identified and all had a gross appearance and histological architecture typical of the teleost eye (Figure 1).5,6 The fatty choroid was grossly apparent, and histologically was composed mostly of adipocytes; choroidal fat was roughly 25% of the A-P volume of the globe in a single small specimen, and roughly 50% in larger adult fish. The choroidal rete was located entirely within the fatty region and dorsal to the optic nerve, as seen via imaging. Meridionally oriented blood vessels within the anterior 25% of the fatty region fed the choriocapillaris at the inner limit of the choroid, just posterior to the retina. The interface between the fatty choroid and the inner sclera was lined by a thin band of connective tissue and pigmented cells. The optic nerve coursed ventrally toward the retina, and in some sections a band of larger-caliber, thin-walled vessels and associated connective tissue paralleled this course. Throughout the optic nerve, inner sclera, and outer choroid were numerous eosinophilic granular cells. While their exact histogenesis and function are unclear, these are considered a normal component in some tissues in fish (e.g., gills) and can be increased in inflammation.4 The presence of eosinophilic granular cells in normal ocular tissues has not previously been described, to our knowledge.
Varying amounts of fat have been noted within the choroid of ocular specimens from other fish species (D. Dubielzig, unpublished data), but this report in the black pacu is the first to carefully document this unique attribute. The function of choroidal adipose tissue is unknown. A role in insulation or heat production seems unlikely in a species with subtropical to tropical distribution. Additional hypotheses to consider include local energy storage, as has been hypothesized for lipid and glycogen inclusions within the choroidal cells of lamprey;2 local storage of fat-soluble nutrients; a role in buoyancy; modification of the optics of the eye; and damping functions against extrinsic stimuli possibly deleterious to the visual process. For the moment, however, the fatty choroid of the black pacu remains an anatomic enigma.
Endnotes
Products mentioned in the text:
a. Philips iE33 linear 11-3 MHz and curvilinear 9-4 MHz probes, Philips Medical Systems, Andover, MA, USA.
b. GE Lightspeed 16, 2.5 and 0.625 mm slices, GE Healthcare, Princeton, NJ, USA.
c. BioSpec 7T, Bruker BioSpin MRI GmBH, Wein, Austria.
Acknowledgments
The authors thank the Steinhart Aquarium, California Academy of Sciences, for donating the black pacu used in this study.
Literature Cited
1. Barnett, C.H. The structure and function of the choroidal gland of teleostean fish. J Anat. 1951;85(2):113–119.
2. Dickson, D.H. and D.A. Graves. Giant choroidal cells in the lamprey eye (Petromyzon marinus L.). Exp Eye Res. 1981;32(3):301–311.
3. Filho, D.W., C. Giulivi, A. Boveris. Antioxidant defences in marine fish—I. Teleosts. Comp Biochem Physiol C Pharmacol Toxicol Endocrinol. 1993;106(2):409–413.
4. Reite, O.B. and Ø. Evensen. Inflammatory cells of teleostean fish: a review focusing on mast cells/eosinophilic granule cells and rodlet cells. Fish Shellfish Immun. 2006;20(2):192–208.
5. Schwab, I.R., R.R. Dubielzig, C. Schobert. Evolution's Witness: How Eyes Evolved. New York: Oxford University Press; 2012.
6. Walls, G.L. The Vertebrate Eye and its Adaptive Radiation. New York: Hafner Publishing; 1963:1–814.
7. Wittenberg, J.B., B.A. Wittenberg. The choroid rete mirabile of the fish eye. I. Oxygen secretion and structure: Comparison with the swimbladder rete mirabile. Biol Bull. 1974;146(1):116–136.
8. Wittenberg, J.B., R.L. Haedrich. The choroid rete mirabile of the fish eye. II. Distribution and relation to the pseudobranch and to the swimbladder rete mirabile. Biol Bull. 1974;146(1):137–156.