L. Smart
In the setting of shock, rapid fluid administration is often life-saving. Isotonic crystalloids remain the first choice in the majority of patients, with the least amount of evidence for causing harm. However, there is growing discussion that the administration of large volumes of crystalloid fluid may cause harm, especially for patients with systemic inflammation and increased vascular permeability. This talk will outline the potential adverse effects of large volumes of crystalloid, including those related to the constituents of the fluid (chloride, acetate), wash-out of the endothelial glycocalyx, and pro-inflammatory responses.
Constituents of Isotonic Crystalloids
The adverse effects of using 0.9% saline are probably the most well-recognised detriment of using high volumes of crystalloid fluid. The two main concerns with using 0.9% saline for blood volume expansion are dilutional acidosis and acute kidney injury. Dilutional acidosis is created by delivery of excessive chloride and subsequent dilution of bicarbonate, an effect that has been documented in dogs with haemorrhagic shock.1 However, in this study, the change in blood pH was small and self-limiting. Still, if a patient is already severely acidaemic, reducing the blood pH further may have adverse consequences. The second concern is increasing the risk of acute kidney injury; which has been raised as a potential in human medicine.2 The theory is that delivery of large amounts of chloride to the macula densa may reduce much-needed renin-angiotensin-aldosterone system activation in times of need (i.e., reduced renal perfusion). It is unknown if this effect is clinically relevant for veterinary medicine but suffice to say, 0.9% saline can be easily avoided and should only be used in alkalaemic patients.
The remaining choices for isotonic crystalloid administration belong in the group of ‘balanced’ fluids, which avoid excessive chloride by providing other anions such as lactate, gluconate and acetate. The most commonly used fluid is lactated Ringer’s solution or compound sodium lactate. The provision of lactate does not contribute to lactic acidosis but provides a substrate for bicarbonate production. However, in a patient that can’t metabolise lactate due to liver dysfunction, this fluid may cause mild hyperlactataemia. This is only inconvenient in the sense that it is difficult to then use lactate as a marker of perfusion. The remaining choices are fluids containing combinations of acetate and gluconate, such as Plasmalyte-148. These anions are metabolised by the kidneys and skeletal muscle, providing a source of buffer. One possible adverse effect of injecting acetate rapidly is vasodilation,3 which may momentarily worsen perfusion. Also, acetate has been shown in rats to be somewhat pro-inflammatory when used for fluid resuscitation.4,5 Therefore, if a choice can be made between a balanced crystalloid with either lactate or acetate, then the fluid with lactate is probably the better choice.
Shedding of the Endothelial Glycocalyx
The endothelial glycocalyx (EG) is a carbohydrate-rich scaffold of proteoglycans and glycosaminoglycans on the luminal surface of endothelial cells (Figure 1). The meshwork of carbohydrates and proteins creates an immobile plasma layer and barrier, preventing interaction between cells in circulation and endothelial cells. This prevents adhesion of platelets and white blood cells, and discourages thrombosis.
| Figure 1. The main components of the endothelial glycocalyx | 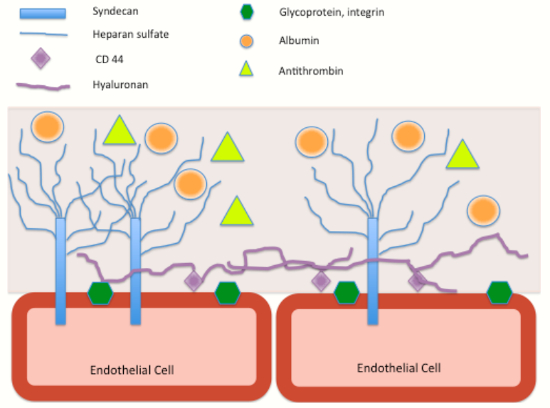
|
|
| |
The EG also plays an important role in fluid flux across the endothelium, which has led to a revision of the traditional Starling’s forces.6 Albumin molecules within the EG layer provides some oncotic pressure, and it is the oncotic balance between the intravascular (flowing) compartment and the EG compartment that affects fluid flux according to protein levels. This explains why raising oncotic pressure in the intravascular compartment does not actually draw fluid from the interstitial space, much to the disappointment of clinicians attempting to reduce peripheral oedema.
It is likely that shedding of the EG is one of the first steps in inflammation and coagulation, which then allows interactions between endothelial cells and circulating leucocytes and platelets.7-11 There is some evidence that rapid intravenous crystalloid fluid therapy, without coinciding injury or inflammation, can increase EG shedding.12 It is unknown if this response to blood volume expansion is related shear stress, release of atrial natriuretic peptide, dilution of EG constituents, or a combination of these factors. In studies using euvolaemic models, it is difficult to separate the effects of hypervolaemia from the effects of the fluid infusion itself and the scenario is less applicable to emergency medicine. Several rodent haemorrhagic shock models comparing large volumes of isotonic crystalloids to plasma or albumin infusions have demonstrated that large-volume crystalloids are associated with greater glycocalyx shedding, vascular permeability and reduced glycocalyx thickness. Preliminary work conducted by our group found that dogs given Plasmalyte-148 for haemorrhagic shock showed evidence for more glycocalyx shedding and inflammation, compared to those given synthetic colloid fluids or whole blood.13 These types of studies raise cause for concern that large volumes of crystalloid, rapidly administered, may not be innocuous as previously thought, and may be contributing to a pro-inflammatory states that our patients may already be experiencing. Ongoing work is being conducted in this area, especially in the area of sepsis,14 where systemic inflammation and vascular dysfunction may make patients particularly sensitive to further EG shedding.
Are Synthetic Colloid Fluids Safer?
Some may be tempted to use synthetic colloid fluid to avoid the administration of large volumes, which may reduce degree of shear stress and atrial distension (therefore avoiding release of natriuretic peptides). However, in order to achieve the same degree of blood volume expansion, the blood will still be diluted. Therefore, there may still be some adverse effect on the endothelial glycocalyx. Also, the choice of diluent for synthetic colloids may be limited; it is most commonly suspended in 0.9% NaCl, which is not ideal for the reasons discussed above.
Additional adverse effects of synthetic colloids include interference with haemostasis, either due to platelet dysfunction or interference with secondary coagulation. Therefore, in a patient with active bleeding, this may not be the best choice. Also, an association between synthetic colloid fluids and acute kidney injury is currently being investigated.
References
1. McBride D, Raisis AL, Hosgood G, Smart L. Hydroxyethyl starch 130/0.4 compared with 0.9% NaCl administered to greyhounds with haemorrhagic shock. Veterinary Anaesthesia and Analgesia. 2017;44(3):444–451.
2. Yunos NM, Bellomo R, Hegarty C, Story D, Ho L, Bailey M. Association between a chloride-liberal vs chloride-restrictive intravenous fluid administration strategy and kidney injury in critically ill adults. JAMA 2012;308(15):1566–72.
3. Saragoca MA, Mulinari RA, Bessa AM, Draibe SA, Ferreira Filho SR, Ribeiro AB, Ramos OL. Comparison of the hemodynamic effects of sodium acetate in euvolemic dogs and in dogs submitted to hemorrhagic shock. Brazilian Journal of Medical and Biological Research 1986;19(3):455–458.
4. Schimmer RC, Urner M, Voigtsberger S, Booy C, Roth Z’Graggen B, Beck-Schimmer B, Schlapfer M. Inflammatory kidney and liver tissue response to different hydroxyethylstarch (HES) preparations in a rat model of early sepsis. PLoS One 2016;11(3):e0151903.
5. Voigtsberger S, Urner M, Hasler M, Roth Z’Graggen B, Booy C, Spahn DR, Beck-Schimmer B. Modulation of early inflammatory response by different balanced and non-balanced colloids and crystalloids in a rodent model of endotoxemia. PLoS One 2014;9(4):e93863.
6. Woodcock TE, Woodcock TM. Revised Starling equation and the glycocalyx model of transvascular fluid exchange: an improved paradigm for prescribing intravenous fluid therapy. British Journal of Anaesthesia 2012;108(3):384–94.
7. Mulivor AW, Lipowsky HH. Role of glycocalyx in leukocyte-endothelial cell adhesion. American Journal of Physiology Heart and Circulatory Physiology 2002;283(4):H1282–H91.
8. Chappell D, Dorfler N, Jacob M, Rehm M, Welsch U, Conzen P, Becker BF. Glycocalyx protection reduces leukocyte adhesion after ischemia/reperfusion. Shock. 2010;34(2):133–139.
9. Constantinescu AA, Vink H, Spaan JA. Endothelial cell glycocalyx modulates immobilization of leukocytes at the endothelial surface. Arteriosclerosis, Thrombosis, and Vascular Biology 2003;23(9):1541–1547.
10. Pries AR, Secomb TW, Jacobs H, Sperandio M, Osterloh K, Gaehtgens P. Microvascular blood flow resistance: role of endothelial surface layer. American Journal of Physiology 1997;273(42):H2272–H9.
11. Gotte M, Bernfield M, Joussen AM. Increased leukocyte-endothelial interactions in syndecan-1-deficient mice involve heparan sulfate-dependent and independent steps. Current Eye Research 2005;30:417–422.
12. Powell M, Mathru M, Brandon A, Patel R, Frolich M. Assessment of the endothelial glycocalyx disruption in term parturients receiving a fluid bolus before spinal anesthesia: a prospective observational study. International Journal of Obstetric Anesthesia. 2014;23(4):330–334.
13. Smart L, Boyd CJ, Claus MA, Bosio E, Hosgood G, Raisis A. Large-volume crystalloid fluid is associated with increased hyaluronan shedding and inflammation in a hemorrhagic shock model. Critical Care 2018;22(Suppl. 1):P293.
14. Macdonald SPJ, Taylor DM, Keijzers G, Arendts G, Fatovich DM, Kinnear FB, Brown SGA, Bellomo R, Burrows S, Fraser JF, Litton E, Ascencio-Lane JC, Anstey M, McCutcheon D, Smart L, Vlad I, Winearls J, Wibrow B. REstricted Fluid REsuscitation in Sepsis-associated Hypotension (REFRESH): study protocol for a pilot randomised controlled trial. Trials. 2017;18(1):399.