Introduction
Cats with chronic kidney disease (CKD) frequently lose weight or have low body condition score (BCS). Although not yet shown in prospective studies, preservation of body weight and lean body mass (LBM) may enhance survival and quality of life in aging cats and those with CKD. Indeed, some loss of weight is part of normal aging, and is not caused by apparent illness. Nutrition offers the possibility to improve longevity as well as quality of life (QoL).
Sarcopenia, the age-related loss of lean body mass (LBM), is a gradual process: initially it is unapparent because increases in body fat persist, and may even cause increases in body weight. However, the loss of LBM has profound effects on survival. Studies have identified decreased survival associated with thin body condition.1-3
In one study of cats with large cell lymphoma, those that lost >5% body weight during treatment had a significantly shorter survival time than those who were able to maintain their weight.4 Patients with other cancers that had a slightly increased or ideal body condition score (BCS) survived up to six times longer than underweight cats.5 In chronic kidney disease (CKD), cats with higher BCS lived longer than those with lower BCS.6 Similarly, cats with heart failure in ideal or slightly greater BCS fared better compared to cats that were underweight or obese.7
Is it possible to prevent cats with CKD from losing muscle and body condition? Is it possible to prevent cats with CKD from losing muscle and body condition? Several studies have shown a decreased ability to digest fat,8,9 protein,8,10 and micronutrients11 in otherwise healthy aging cats. Because of this, a high energy, highly digestible diet with an increased protein content may be appropriate. Studies in healthy cats suggest that 5–6 g protein/kg body weight is needed to maintain or enhance LBM.12,13 One small, unpublished study in middle-aged cats with mild CKD showed they could maintain LBM over 30 weeks when fed only 6.5 g protein/100 kcal.14 However, it is unclear what the actual protein intake was, as no data on actual food intake or body weight was included (i.e., protein calories ingested). If cats are eating only small amounts of food, a higher percentage of calories from protein may be needed to meet their needs.
Old cats need protein. The majority of cats with CKD live a long time. Given the importance of protein in cats, how do we optimize the benefits of a renal diet but correct or prevent further muscle wasting?
Clinical Implications
Step 1: Evaluate
Just as every cat is different, the needs of different cats with CKD may differ. It is important to start by finding out exactly what, (foods, treats, supplements), and how much, a patient is eating. A simple diet history form can be found at: www.wsava.org/nutrition-toolkit. Evaluating body condition entails not just assigning a body condition score (BCS, Figure 1), but also getting a weight at every visit. Both body weight and BCS are subjective ways to evaluate caloric adequacy: weight reflects the current state and BCS, gives a longer-term perspective.
| FIgure 1 | 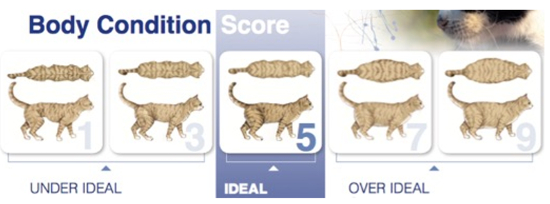
Body Condition Score (from www.wsava.org/WSAVA/media/Arpita-and-Emma-editorial/Body-Condition-Score-Cat.pdf |
|
| |
Calculating the percent weight change is an easy way to follow trends and get a better idea of weight relative to size. This important measure helps alert both the practitioner and the client to insipient (or blatant) physiologic alterations.

Loss of muscle can occur without fat loss or a decrease in BCS and individuals can retain an obese or overweight BCS yet be under muscled. Muscle condition scoring (MCS, Figure 2, Table 1) is a subjective way to evaluate LBM. Therefore, both subjective BCS and MCS should be performed in all cats, to evaluate fat mass and lean mass independently of each other.17 When clients participate in determining BCS, it is more meaningful to them.
Similarly, they can be taught to assess muscle condition.
| Figure 2 | 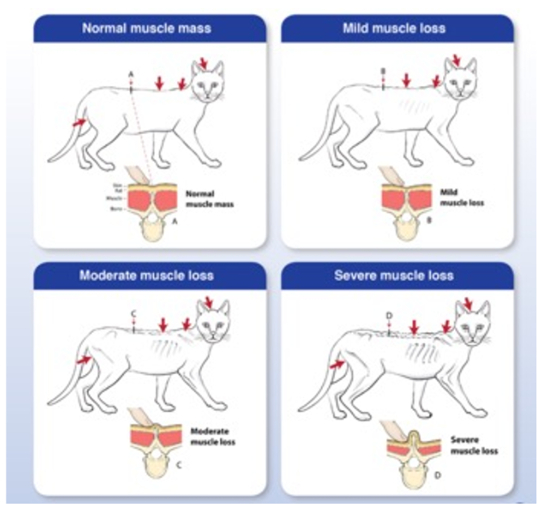
Muscle condition score (from www.wsava.org/WSAVA/media/Documents/Committee%20Resources/Global%20Nutrition%20Committee/Muscle-condition-score-chart-Cats-1.pdf |
|
| |
Table 1. Muscle condition scoring criteria18

Older cats may have difficulty getting to food without discomfort; this is often due to arthritis, but loss of muscle mass and function can be a contributing factor. Weight loss can occur due to reduced access to food or reduced intake due to a decline in the ability to taste, smell, or see food. Pain (commonly from oral disease) and nausea can interfere with eating. Nausea, associated with hypergastrinemia is believed to occur in some cats with CKD. Gastroprotectants or proton pump inhibitors may be beneficial depending on the cause of the gastric upset. Omeprazole has been shown to be superior to H2 antagonists (i.e., ranitidine) in reducing gastric acid production.19
If physical and laboratory evaluations fail to reveal a cause for the weight loss, before attributing this problem solely to old age or before considering medical intervention, the cat’s environment should be considered. What may not be threatening to us, or even to the cat in the past, may be perceived as a source of stress in the older, less confident cat. A discussion about ease of access to all-important resources may find a crucial defect that may be readily corrected.20 A household resource checklist may be downloaded from www.cliniciansbrief.com/article/household-resource-checklist.
To encourage intake, multiple bowls of dry food should be located around the home in safe, quiet, private places. Moist food should be fed 2–4 times per day. Water should be fresh and, like food bowls, should also be in a variety of locations, and not adjacent to food bowls. Treats should be complete and balanced (e.g., Smartbites™). Bowls should be wide to allow whisker clearance (especially in a clinic setting). For some cats with cervical or coxofemoral degenerative joint disease or lumbosacral spondylosis, raising the bowl may be helpful. (Photo 1.)
| Photo 1 | 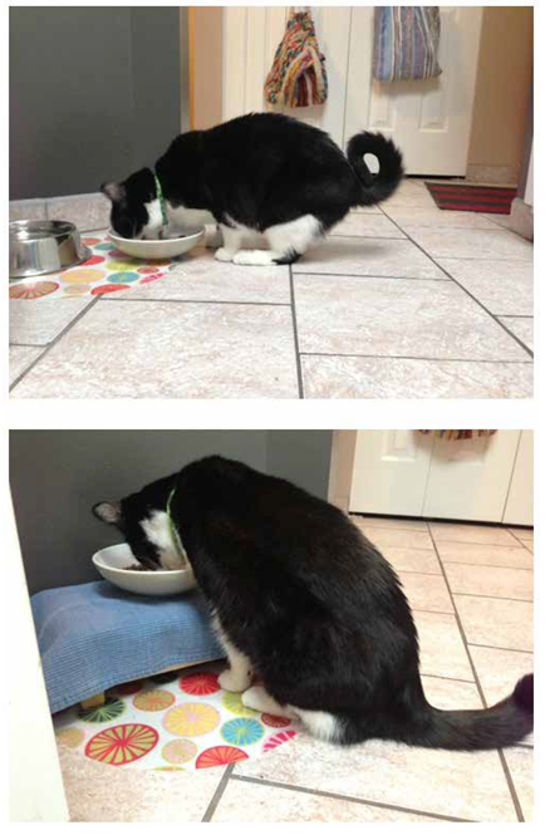
A) Lumbosacral and coxofemoral changes make sitting down uncomfortable. B) Raising food and water bowls may help cats be more comfortable. |
|
| |
Step 2: Calculate
Previous weight, BCS and MCS may suggest the ideal body weight for the individual cat. There are several formulae available but a simpler “rule of thumb” is:
- Intact, active cats need 60–80 kcal/kg ideal weight/day.
- Sexually altered, less active cats need 40–50 kcal/kg ideal weight/day.
- Older cats (>12 years) need 70 kcal/kg ideal weight/day.
Using the information regarding what the patient is currently eating, along with physical examination findings, (including hydration), provides a starting point for a nutritional plan. Both the weight loss and muscle wasting need to be addressed.
Caloric requirements should be calculated in order to determine the amount of food required. Ideally, this should be based on a good diet history with known intake as individual calorie requirements can differ greatly from calculated averages. If fed dry food, it should be available at all times, in quiet locations that are readily accessed by this individual. If a cat prefers to eat only a small quantity at a time, moist food may not be appropriate as it is less calorically dense than dry food. While moist food may be preferable in providing more moisture, some cats are reluctant to eat it.
When recommending a change in the diet, recommend that the client monitor closely the amount eaten and make the change gradually over 3 or more weeks. It is not imperative that the cat switches completely to the new food. Within reason, it is more important that they eat adequate amounts rather than what they eat.
Example:
- 4.0 kg cat BCS 3/9, moderate muscle wasting who previously weighed 4.5 kg BCS 5/9 good muscle mass.
- At 70 kcal/kg/day, this cat needs 315 kcal/day for 4.5 kg ideal weight.
- Protein requirement is at least 5 g/kg ideal weight/day: this cat needs >23 g protein/day.
Example using a dry or canned renal diet:
- Let’s say the dry formulation has 3936 kcal/kg (3.94 kcal/g) or 398 kcal/8 oz cup can as fed.
- It has 6.61 g protein/100 kcal.
- For a 4.5 kg cat who needs 315 kcal/day this equals 80 g or 4/5th of a cup of this diet/day.
- For 315 kcal @ 6.61 g protein/100 kcal there would be 21 g protein in the day’s dry. This is less than the minimum of 23 g/day needed to maintain muscle condition.
- For the same diet, the canned has 193 kcal/5.5 oz can, therefore, 315 kcal = 1.6 cans.
- It has 7.23 g of protein/100 kcal. For 315 kcal @ 7.23 g protein/100 kcal there would be 23 g protein in the day’s canned food.
It is important that the cat has a good appetite and eats the entire quantity of dry or canned food. With lower calorie intake, protein intake will be deficient. A renal diet has benefits, (i.e., low phosphorus, buffering agents, +/- n-3 fatty acids, etc.), but should the response to the diet reveal inadequate muscle mass, then one needs to supplement the diet with protein or change the diet and use an intestinal phosphate binder, in order to both meet the cat’s nutritional needs while still addressing the CKD.
Step 3: Communicate
Once the ideal body weight has been estimated, either based on historical information or on an educated guess, and the calories needed to maintain this ideal weight are calculated, it should be noted in the medical record. Regardless of diet chosen, the daily quantity of food (dry and or moist) should be calculated and recorded. The amount of food (grams, cups, cans) that the cat needs to eat must be conveyed to the client both verbally and in writing. Also, make sure that they understand that this is a starting point. For the sake of simplicity, and because the cat and the client may have preferences, the author likes to send home samples of each recommended diet for the cat to try. Once the cat has made their (initial) choice, the quantities can be calculated and given to the client. Do not try to introduce the new diet in the clinic: a vigilant cat is unlikely to try or like a new diet. Additionally, they may develop a diet aversion.
Step 4: Reassess
It is important to recheck patients 1–2 weeks after making nutritional recommendations to assess the effect of the food on the cat’s condition. This is no different than reassessing the impact of other medical recommendations, (e.g., medications, fluids, etc.), or repeating laboratory tests. It is not uncommon that a patient’s decline or lack of improvement is because of feeding factors rather than an inherent progression of their illness. Determine whether the cat is eating enough of the recommended food to result in an improvement in their weight, BCS, and MCS. Find out how the client feels about the diet, feeding the diet, the cat’s enjoyment, and QoL on this diet. Providing nutrients may be the most challenging part of any therapeutic regimen, and recovery or attaining the best possible QoL in cats may depend on our ability to ensure optimal nutrition. Again, communication with, and motivation of, the client is crucial.
Step 5: Upping the Ante
If body weight, condition and MCS are not improving, additional intervention is required. Verify that the diet appeals to the cat, taking into account its texture, aroma, taste, and consistency. Confirm that the cat is able to hide when they feel the need and has a safe, private places to eat. Feline facial pheromone may be beneficial to reduce stress.20 Verify that inadequate food intake isn’t due to other factors, such as dysphagia, pain (e.g., oral, dental, gastrointestinal, multisystemic, etc.), dislike of the diet (e.g., boredom, altered palatability, spoilage), aversion, fear (e.g., environmental changes including those in the social demographics). Palatability may be improved by bringing the temperature of moist food to body temperature and by changing the consistency of the diet.
Rehydration and correction of electrolyte imbalances is important but may have been overlooked or the need may have changed. Anti-emetics (e.g., maropitant, mirtazapine, dolasetron, ondansetron) have a place if the cat is vomiting.
Appetite stimulants including cyproheptadine (1 mg/cat PO BID), mirtazapine (1–2 mg/cat PO q48h)21 may help to jump-start a cat’s appetite, but it is important to keep track of total calories consumed. A cat eating small amounts of baby food will not meet their caloric needs: they need 2–3 jars/day. Meat baby food is not balanced, but is sufficient for several weeks. If a cat is eating, but not enough, supportive feeding (assisted syringe feeding or tube feeding) must be considered. There are several diets specifically designed for the assisted feeding of cats (e.g., Purina® Pro Plan® Veterinary Diets CN Critical Nutrition™), liquid balanced enteral diets for cats. Additionally, we can make a slurry from any canned food; blend with a liquid feline diet rather than water to minimize loss of calories. If the weight is improving, but MCS is not, supplementing with 1 oz (28 g) of cooked chicken/day may be a low phosphorus protein option.
If the cat is reluctant to eat the renal diet or the MCS is not improving, consider feeding the diet that cat prefers and utilizing intestinal phosphate binders. Aluminum hydroxide dry gel powder (USP) is well accepted by most cats and can be mixed directly into moist food or added to dry. (Put the AlOH dry gel powder and dry food together in a plastic bag, shake.) (www.zzcat.com/CRF/supplies/binders.htm)
Step 6: Supportive Feeding
There are several options for assisted feeding each with advantages and disadvantages. Syringe feeding, nasogastric (NG), esophagostomy, or gastrotomy tubes are the most common choices. In general, the author starts with syringe assisted feeding until the cat is stable enough to allow the brief anaesthetic required for the placement of an esophageal tube. Syringe feeding can be very successful bearing a few things in mind. Because the oral capacity of a cat is only 1/2–1 ml, small volume syringes should be used being sure to provide the entire caloric dose. Like an NG tube, syringe feeding is suitable only for the short-term. A large bore feeding tube should be considered early and as a temporary measure to improve the nutritional plane (BCS, MCS) of the patient with kidney disease.
In cats with concurrent liver disease, three doses of Vitamin K1 (1.0 mg/kg q12h SC) should be given prior to tube placement, biopsies or any other procedure that might result in bleeding. Placement of esophageal tubes is not complicated and details are discussed elsewhere. (An example is listed in the resource below.) Instrumentation is very basic requiring only the following: 14–16 Fr red rubber feeding tube/urinary catheter, long curved forceps, a scalpel blade, suture and bandaging materials (or a KittyKollar™), and a multiple use injection port (“prn adaptor”).
Calculating how much to feed requires that you know the calories they need to maintain their ideal, healthy weight as well as the caloric densities (kcal/ml) of the diet you are intending to use (see Table 2). Start by feeding 1/3 of the calories needed, on day two, feed 2/3, and on day three, feed the full calories needed for the ideal weight.
Table 2. Caloric densities of convalescent diets, for calculating feeding volumes
|
Clinicare™: 1 kcal/ml
|
|
Royal Canin/MediCal Recovery™: 1.04 kcal/ml
|
|
Hill’s a/d™: 1.17 kcal/ml
|
|
Purina PPVD CN™: 1.33 kcal/ml
|
|
Blending a renal (or any other) diet with a liquid diet will provide a higher caloric density than if water is used.
|
Example
- 4.0 kg sick cat BCS 3/9, healthy weight 4.5 kg BCS 5/9 Using the calculator, 216 kcal by day 3.
- 315 kcal = 302 ml of Royal Canin Recovery™; 269 ml of Hill’s a/d™.
- 236 ml of Purina PPVD CN™.
Example, using PPVD CN, the most calorically dense:
- Day 1 feed 80 ml.
- Day 2 feed 160 ml.
- Day 3 feed 236 ml.
With surgically placed tubes there is a delay in how quickly one can start to use them; with an esophageal tube only a 2–3-hour delay is required to ensure full recovery from anaesthesia. Cats can eat with the tube in place although it is recommended to avoid offering food for the first week to reduce the likelihood of them developing an aversion to the food offered. Once a cat is eating well with tube in place the question becomes when one can remove the tube. Weigh the cat and, as long as they are eating well, avoid using the tube (medications are okay) for a week then reweigh them. If the weight is stable (or increased), then it is safe to remove the tube. Because of stoma formation, removal does not require anaesthesia. Remove the suture (purse-string or stay sutures) and pull the tube out. Suturing is not required for the skin opening. Cleanse minimal serous discharge that may occur for 2–3 days.
Feeding frequency: the number of feedings per day, (and hence intervals), is determined based on the volume of food tolerated per feeding. Start with 6 ml and increase by 6 ml increments to about 36–48/feeding for most cats. In the uncommon case of the patient who cannot tolerate even 6 ml boluses despite antiemetic therapy, trickle feeding may be instituted. Trickle feeding is a technique in which liquefied food is syringed into an empty fluid bag and administered gravitationally or by pump assistance via an intravenous line attached to the large bore feeding tube or by use of a large syringe filled with food and syringe pump. Renew food and delivery tubing and syringe at 12-hour intervals to avoid bacterial contamination. A promotility agent may be warranted as well.
The success of assisted feeding is measured objectively by weight gain. Subjective measures will include improved coat quality, increased energy, muscle recovery, and innumerable other effects that the client will appreciate. An improved QoL is the goal whether recovery from the underlying problem is possible or not.
Summary
Renal diets provide benefit for cats with CKD. It is imperative that the cats eat enough calories and protein to optimize their body and muscle condition scores. Each patient must be assessed individually and monitored.
This requires client communication and recheck visits. Cats with chronic kidney disease with higher BCS will live longer, and have an improved quality of life.
Useful Resource
Hodshon B, Tobias K. Esophagostomy feeding tubes, Clinicians Brief. February 2014 (www.cliniciansbrief.com/article/esophagostomy-feeding-tubes)
References
1. Scarlett JM, Donoghue S. Associations between body condition and disease in cats. J Am Vet Med Assoc. 1998;212:1725–1731.
2. Doria-Rose VP, Scarlett JM. Mortality rates and causes of death among emaciated cats. J Am Vet Med Assoc. 2000;216:347–351.
3. Freeman LM. Cachexia and sarcopenia: emerging syndromes of importance in dogs and cats. J Vet Intern Med. 2012;26:3–17.
4. Krick EL, Moore RH, Cohen RB, et al. Prognostic significance of weight changes during treatment for feline lymphoma. J Fel Med Surg. 2011;13:976–983.
5. Baez JL, Michel KE, Sorenmo K, et al. A prospective investigation of the prevalence and prognostic significance of weight loss and changes in body condition in feline cancer patients. J Fel Med Surg. 2007;9:411–417.
6. Freeman LM, Lachaud MP, Matthews S, et al. Evaluation of weight loss over time in cats with chronic kidney disease. J Vet Intern Med. 2016;30(5):1661–1666.
7. Finn E, Freeman LM, Rush JE, et al. The relationship between body weight, body condition, and survival in cats with heart failure. J Vet Intern Med. 2010;24:1369–1374.
8. Perez-Camargo G. Cat Nutrition: what’s new in the old? Compend Contin Edu Pract Vet. 2004 26(Suppl 2A):5–10.
9. Perez-Camargo G, Young L. Nutrient digestibility in old versus young cats. Compendium. 2005;27(3A):84.
10. Bermingham EN, Weidgraaf K, Hekman M, et al. Seasonal and age effects on energy requirements in domestic short-hair cats (Felis catus) in a temperate environment. J Anim Physiol Anim Nutr. 2012;97:522–530.
11. Perez-Camargo G, Young L. Nutrient digestibility in old versus young cats. Compendium. 2005;27(3A):84.
12. Nguyen P, Leray V, Dumon H, et al. High protein intake affects lean body mass but not energy expenditure in nonobese neutered cats. J Nutr. 2004;134:2084S–2086S.
13. Laflamme DP, Hannah SS. Discrepancy between use of lean body mass or nitrogen balance to determine protein requirements for adult cats. J Feline Med Surg. 2013;15:691–697.
14. Yu S, Velliquette R, Yamka R, et al. Dietary crude protein of 28.5% maintains long-term lean body mass in cats with impaired kidney function. J Vet Intern Med. 2011;25(3):734. (Abstr NM-9).
15. Laflamme DP. Loss of lean body mass in aging cats is affected by age and diet. European Society Veterinary & Comparative Nutrition, Annual Conference. Ghent, Belgium; Sept 19–21:2013 (Abstr).
16. Wolfe RR. The underappreciated role of muscle in health and disease. Am J Clin Nutr. 2006;84:475–482.
17. Freeman L, Becvarova I, Cave N, et al. WSAVA nutritional assessment guidelines. J Fel Med Surg. 2011;13:516–525.
18. Michel KE, Anderson W, Cupp C, et al. Correlation of a feline muscle mass score with body composition determined by DEXA. Brit J Nutr. 2011;106:S57–59.
19. Šutalo S, Ruetten M, Hartnack S, et al. The effect of orally administered ranitidine and once-daily or twice-daily orally administered omeprazole on intragastric pH in cats. J Vet Intern Med. 2015;29:840–846.
20. Ellis SLH, Rodan I, Carney HC, et al. AAFP and ISFM feline environmental needs guidelines. J Feline Med Surg. 2013:15:219–230.
21. Quimby JM, Lunn KF. Mirtazapine as an appetite stimulant and anti-emetic in cats with chronic kidney disease: a masked placebo-controlled crossover clinical trial. Vet J. 2013;197:651–655.