Elbow dysplasia has been found in 78 breeds evaluated by the Orthopedic Foundation for Animals, which opened its ED database in 1990. The incidence of elbow dysplasia in these breeds ranged from 1.2 to 47.9 percent of the evaluated dogs.
Elbow dysplasia can lead to lameness or abnormal gait, but a number of affected dogs show no obvious clinical manifestations. Three factors produce elbow dysplasia, either singularly or in any combination.
Elbow dysplasia can be extremely debilitating, but there is no satisfactory medical protocol or surgical procedure that can significantly alter the progression of the disorder or cure it. This makes it increasingly important to reduce the incidence of the disease through selective breeding, which has been shown to reduce its incidence.
The terminology of elbow dysplasia (ED) was introduced in 1961 to describe a generalized osteoarthrosis (OA) of the elbow reported in association with an ununited anconeal process (UAP). A report followed that revealed the same OA
without the UAP. Over time, fragmentation of the medial coronoid process (FCP) of the ulna and osteochondrosis dissecans (OCD) of the humeral condyle also were described with this generalized elbow arthritis. These three components are the currently-accepted entities comprising elbow dysplasia.
UAP, FCP and OCD may be present singularly or in any combination. It has been reported that FCP with OCD can occur with a frequency as high as 37 percent . With such a high frequency the question arises whether the second lesion is a result of the first lesion in contrast to the two lesions being present as separate entities. Certain breeds tend to be affected with a particular entity more frequently than the other components, which lends support to the heritability of the disorder.
Etiology
The exact mechanism of these abnormalities has not been clearly defined.
There are two different theories for the resulting lesions. The first theory, proposed by Olsson, was that all three disorders are manifestations of osteochondrosis. Osteochondrosis is a disturbance of endochondral ossification, which is the formation of bone through the ossification of cartilage. In the area of the abnormality, there is a thickening of the cartilage due to deprivation of nutrients supplied to the chondrocytes by diffusion from the synovial fluid. The cells at the bottom of the thickened area do not receive adequate nutrition and become necrotic, hence the cartilage in this area will not be attached to the underlying bone. Movement of the bones in the joint provide the forces necessary to break this thickened area free, forming a cartilage flap or a complete full-thickness cartilagenous defect.
| 
Elbow dysplasia can be an extremely debilitating disorder for which there are no satisfactory treatments. Prevention, through selective breeding, is the best means of management. |
|
| |
The movement of the bony or cartilaginous fragments prevents healing of the exposed subchondral bone. Pain resulting in lameness, due to inflamed nerve endings in the subchondral bone, may persist or may diminish with time. The fragments serve as a constant irritant, exacerbating lameness and the progression of arthritic changes. This theory was supported by the different components of elbow dysplasia present in the elbows of the same animal.
The second theory, proposed by Wind, is of joint incongruity as a primary cause of the fragmentation or resulting lack of fusion (ununited). Congruency is important in the elbow joint because three bones must fit together smoothly to allow for a gliding movement in flexion and extension as well as internal and external rotation.
The entire ulnar trochlear notch is in close contact with the articular cartilage of the olecranon fossa and the humeral trochlea which articulates with both the radius and the ulna. Any asynchronous growth of the radius and ulna or insufficient development of the ulnar trochlear notch would result in loading forces in the area of the anconeal process and or the coronoid process. It has been proposed, in two different studies, that these separate components are entirely different disease entities since inheritance is independent.
Presentation
A number of affected dogs show no obvious clinical manifestations of the disorder. Some affected dogs may be clinically lame or have an abnormal gait. In dogs that are clinically lame, varying degrees of lameness maybe exhibited and
exacerbation with activity is seen. Gait abnormality usually is present in dogs with bilateral disease, unless one elbow is worse than the other, making a unilateral lameness evident.
Affected limbs are usually rotated inward with elbows rotated outward. Manipulation of the elbow will reveal a decrease in range of motion. Crepitation, joint effusion, joint capsule thickening and muscle atrophy are variable. This variability is the result of the tremendous range of the abnormality even within a single entity
The radiographic signs of ED and the clinical presentation do not necessarily correlate directly. A dog may have significant radiographic changes and not be clinically lame. In one study, dogs were evaluated when an acute lameness was
present, but no radiographic changes were seen at this time. These same dogs were evaluated at a later date when the clinical lameness had resolved, but radiographic signs of ED were now seen.
This resolution in lameness with a progression of degenerative changes can be attributed to pain in the acute phase of the disease. As healing occurred, the lameness resolved. The damage persisted and, being consistent with the
chronic progressive nature of arthritis, resulted in the subsequent radiographic changes.
Diagnosis
In the young dog with lameness from elbow dysplasia, diagnosis is made from typical clinical signs and standard radiographic evaluation. For diagnosis of a clinically lame patient, views should include a lateral, a craniocaudal and a flexed lateral of the elbow joint. The radiographic appearance is characterized by incongruity and or degenerative changes.
The anconeal process may not fuse to the diaphysis of the ulna until 4 to 5 months of age, so a diagnosis of UAP prior to 4 to 5 months of age is premature. No studies have been conducted for the giant breeds to determine an age range;
this age range maybe even older in the giant breeds. The usual age of presentation with clinical signs is 6 to 12 months, but affected dogs may not be clinically lame until much older.
UAP is bilateral in 20 to 35 percent of affected dogs. Males are more frequently affected than females, potentially because of a more rapid growth rate and a greater over-all size.
The most significant radiographic sign of UAP is the appearance of a radiolucent line of separation between the olecranon and the anconeal process (Figure 1). This line may be present in varying degrees since the anconeal process can actually be partially fused (Figure 2).
Click on an image to see a larger view.
|
| Figure 1. Extreme flexed lateral radiographic projection of the elbow joint. The ununited anconeal process is completely separated from the ulna by the line at the point of the arrow. |
|
| Figure 2. Extreme flexed lateral radiographic projection of the elbow joint. A partially fused anconeal process is evident. The anconeal process is fused caudally and the line at the point of the arrow is the area where fusion failed to occur. |
It is important to obtain a lateral radiograph of the elbow in extreme flexion. If not flexed, the distal, caudal aspect of the medial epicondyle of the humerus is superimposed on the anconeal process, which can be confused with a separation line.
As the disease progresses, sclerosis can be seen along the margins of separation. Degenerative changes subsequently appear at 7 to 8 months of age.
The fragmented coronoid process occurs at 4 to 10 months of age. Unlike a UAP, an FCP may not be visible radiographically (Figure 3). Fissures in the cartilage or slightly irregular articular margins would be the least abnormal
(Figures 4 & 5). These changes are not evident radiographically, but the resulting degenerative changes would eventually become evident radiographically.
Click on an image to see a larger view.
|
| Figure 3. Extreme flexed lateral radiographic projection of the elbow joint. Although not typically evident via radiographic examination, the arrow marks the line of separation of a fragmented medial coronoid process. A small, triangular, radiodense fragment can be seen adjacent to the line of separation. |
|
| Figure 4. A gross tissue specimen of the articular surface of the radius (right) and ulna (left). The arrow points to a fissure in the cartilage. The arrowhead points to an area of roughened articular cartilage. These lesions would not be evident radiographically but could result in changes consistent with Grade 1 elbow dysplasia. |
|
| Figure 5. A gross tissue specimen of the articular surface of the radius (right) and ulna (left). The arrow points to the irregular articular margin between the radius and ulna. It appears that a portion of the ulna is missing (in the proximity of the coronoid process). This lesion would not be evident radiographically, but could result in changes consistent with Grade 1 elbow dysplasia. |
Definitive radiographic identification of FCP often is not possible because of super-imposition of the radial head over the medial coronoid process. In addition, the central radiographic beam rarely intersects the cleavage line in a parallel manner.
The diagnosis of the FCP is typically from the secondary degenerative changes that result from the abnormality (Figures 9 & 10). These are seen as early as 7 to 8 months of age but may not be evident until maturity. These are osteophytes located on the proximal and lateral aspects of the anconeal process. Similar changes on the medial humeral epicondyle and medial aspects of the joint develop as the disorder progresses. Sclerosis between the proximal radius and ulna and/or an increased humeroradial joint space maybe seen on a lateral projection.
The primary radiographic sign of OCD is common to all locations. There is evidence of a subchondral bone defect that causes a flattening or concavity of the articular surface. In the elbow OCD is diagnosed by visualization of either a subchondral bone defect or cartilage flap on the medial humeral condyle. This defect may have a sclerotic margin.
The cartilage flap may become calcified or contain a piece of subchondral bone facilitating recognition radiographically as a "joint mouse."
A calcified cartilaginous flap maybe seen radiographically as a thin, linear, mineral opacity covering the defect. The disease occurs between 4 to 10 months of age. OCD can affect multiple joints (elbow, shoulder, stifle, hock) and is commonly bilateral. If a dog is lame and elbow dysplasia is a suspected diagnosis, then the contralateral elbow should be radiographed as well. A craniocaudal projection or a slightly oblique lateral craniocaudal projection is necessary to visualize a lesion in the elbow (Figure 6).
Click on an image to see a larger view.
|
| Figure 6. Slightly oblique craniocaudal radiographic project of the elbow joint. The arrows emphasize a radiolucent subchondral bone defect from osteochondrosis. |
In general, the amount of arthritic changes that result from elbow dysplasia parallels the amount of joint instability created by the abnormality. Since the UAP is typically the most unstable, it is likewise associated with the most degenerative changes and subsequently the worse prognosis in reference to debilitation. The FCP is less unstable.
The osteochondrosis lesions may not create gross instability but are associated with incongruency and inflammation that would result in degenerative changes.
At this time there have been no scientific studies that have correlated the amount of degenerative change with the amount of dysfunction or prognosis. With the chronic progressive nature of arthritis, it is generally a matter of time before the animal becomes clinically lame from the disorder.
Therapy
Treatment for elbow dysplasia will vary according to the individual case. Factors to be considered are the present amount of degenerative joint disease, age of the patient and degree of lameness. Typically all immature dogs showing
lameness referable to FCP, OCD or a UAP are surgical candidates. Mature dogs with mild to moderate arthritic changes and a component of instability also may be considered for surgery.
Removal of the unstable component may provide for some decrease in pain. Dogs with pain from severe degenerative changes, but in which the joint is stable, are not considered surgical candidates because surgery may disrupt
stability and aggravate the problem. Surgery is recommended to facilitate healing of the cartilagenous defect and to remove the loose fragments.
Depending on the component of elbow dysplasia and the amount of abnormality present at the time of surgery, owners should be aware that arthritis is a progressive disorder and improvement may be seen, but normality probably will not be
achieved. Medical and surgical management is often unrewarding. Fewer than 50 percent of the dogs treated medically and fewer than 60 percent of the dogs treated surgically (for FCP) had a satisfactory long-term recovery.
The rate and extent of arthritic changes are variable. Elbow dysplasia can be a crippling disorder. The degree of osteoarthritis may be severe enough that by the age of 2 years clinical signs are evident and the use of analgesics are indicated for pain relief.
Often the best results (for future use and minimal arthritic changes) are achieved with early intervention. Therefore, a more aggressive surgical approach is advocated. Although some animals do recover spontaneously, this can happen
only if the flap breaks loose and is absorbed in the joint cavity. This process may take 9 to 12 months and bilaterally affected animals (incidence of 27 percent and 68 percent) are unlikely to recover to the point of clinical soundness.
When an animal is markedly lame in one leg, it is difficult to assess lameness in the contralateral leg. Both limbs should be radiographed even if only one limb is clinically lame. An additional danger is that the loose cartilage flap may survive within the joint to become a large ossicle (joint mouse) that will cause severe inflammatory changes and degenerative joint disease. If the flap never breaks free, a similar deterioration of the joint occurs. Partially attached flaps have been removed in 3-year-old dogs.
The best method for surgery would be through arthroscopy by a surgeon who has a significant amount of experience performing arthroscopies, since there is a learning curve associated with this method.
Genetic Databases
Multiple studies support the theory that the various components of ED are heritable. The heritability index and incidence varies by the breed, component and population studied. It appears that the disorder is inherited polygenically with development being multifactorial. Both environmental factors and the additive effect of many genes contribute to expression.
The International Elbow Working Group (IEWG), a consortium of experts from around the world, was founded in 1989 to lower the incidence and prevalence of elbow dysplasia by coordinating worldwide efforts. These efforts include research,
dissemination of information, formulation of guidelines for national registries and provision of education about elbow arthrosis.
It was necessary to develop a protocol for screening elbows that would be acceptable to the scientific community and breeders. It was agreed that ED was the manifestation of inherited FCP, UAP, OCD, articular cartilage anomaly and/or joint incongruity that resulted in elbow arthrosis. Until a DNA test is available for the detection of animals genetically predisposed for ED, genotype can only be estimated by knowledge of the evaluations of the extended family. The IEWG has continued to meet periodically to provide international discussions for the purpose of exchanging information and reviewing
the elbow evaluation protocol.
The OFA started its elbow database in 1990 using a modified protocol of the IEWG. Initially the database was
semi-closed, but since July 1, 2000, owners have had the choice of an open database as well. When establishing criteria for a protocol for screening breeding animals, several aspects need to be considered. To encourage submission of data from
multiple-dog owners (i.e., breeders) the cost must be reasonable. This should take into consideration the cost to the submitters of obtaining such data prior to the entry into the database.
The minimum age for certification must allow for detection of a significant number of affected dogs by that age and still allow for entry of an individual into the breeding program in a timely manner. Therefore, the OFA requires one view of each elbow in extreme flexion. Certification can be obtained at 24 months of age. The age of 2 years is well past the active stage of the disorders and arthritic changes should be evident on the lateral projection, regardless of the magnitude of the abnormality.
The OFA reports elbows as normal (Figure 7) or dysplastic. The abnormals are graded as grades 1 (Figures 8 & 9 through 3 (Figures 10), with grade 1 being the least abnormal. The grades are differentiated by the relative amount of osteophyte formation on the anconeal process. The grades of dysplasia will facilitate the calculation of heritability.
Click on an image to see a larger view.
|
| Figure 7. Extreme flexed lateral radiographic projection of a normal elbow joint. |
|
| Figure 8. Extreme lateral radiographic projection of the elbow joint. The arrow shows the osteophyte development on the anconeal process. The elbow is an example of Grade 1 elbow dysplasia. |
|
| Figure 9. Craniocaudal radiographic projection of the elbow joint. The arrow shows an osteophyte on the proximal ulna. This is an example of Grade 1 elbow dysplasia, but is not a required view for the OFA. |
|
| Figure 10. Extreme flexed lateral radiographic projection of the elbow joint. The arrows show osteophyte development in the area of the anconeal process on the ulna, the caudal aspect of the humerus and the cranial margin of the radius. This is an example of a Grade 3 elbow dysplasia. |
Summary
Elbow dysplasia can be extremely debilitating. The onset is early and patients are bilaterally affected in a significant number of cases. Despite the ability to treat affected dogs, there is no satisfactory medical protocol or surgical
procedure to significantly alter the progression or cure the disorder. This poor response to medical and surgical management is substantiated with force plate gait analysis pre- and post-therapy. This makes it increasingly important to reduce the incidence
of the disease through selective breeding. Selectively breeding phenotypically normalindividuals has been shown to reduce the
incidence of the disorder.
Click on an image to see a larger view.
| The Canine Elbow | 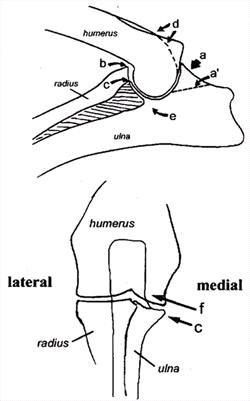
|
|
| |
| The Canine Elbow
a) Anconeal process, site of osteophyte development
a') Line of separation for UAP
b) Site of osteophyte development
c) Medial coronoid process
d) Site of osteophyte development
e) Trochlear notch
f) Site of osteochondrosis lesion |
| Normal versus Dysplastic Elbows, 1989 - 2004 | 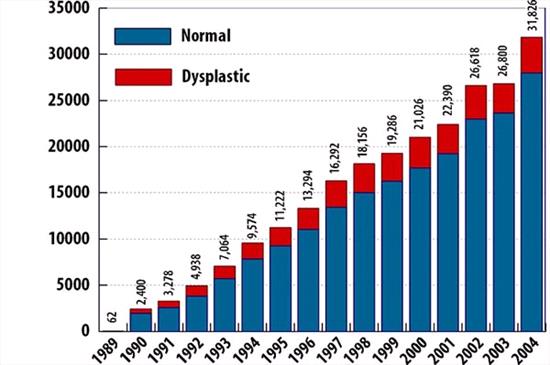
OFA's Elbow Dysplasia Database
From 1989 through 2004, 235,048 dogs were submitted to the OFA elbow database for evaluation. 234,958 were found to be normal. 35,790 dogs (15.23%) were found to be dysplastic. 732 dogs had equivocal results (not represented in this chart). In 2004, 3,854 of the 31,848 dogs tested were found to be dysplastic, 12.10%. |
|
| |