The Queen Mother Hospital for Animals, The Royal Veterinary College, North Mymms, Herts, UK
Notes supplied by Nadja Sigrist, DrMedVet, FVH(Small Animals), DACVECC.
Introduction
Blood gas analysis allows interpretation of acid-base status as well as respiratory function, including both oxygenation and respiration. This lecture focuses on the practical approach of blood gas analysis and refers to the literature for a more in-depth discussion of acid-base balance and (patho)physiology of respiratory function.
Sample Taking
Both arterial and (central) venous blood samples can be used to interpret acid-base status and ventilation, but only arterial blood gases will provide true information regarding oxygenation. Arterial blood gas samples can be derived from any superficial artery, but commonly the femoral artery or the dorsal pedal artery are used. If continuous arterial blood monitoring is required, an arterial catheter in the dorsal pedal artery is more convenient. Arterial samples should be collected easily (without excessive negative pressure or significant venous stasis) and without air contamination and should ideally be analysed immediately. If not they may be kept at 4°C (ice water) for a maximum of 2 hours.
Normal Values
Normal values for blood gas results have been published (Figure 1); however, validated normal ranges of each individual blood gas machine should be used whenever available.
Figure 1. Normal blood gas values in dogs and cats.
|
Parameter
|
Arterial
|
Jugular venous
|
Cephalic venous
|
|
|
Dog
|
Cat
|
Dog
|
Cat
|
Dog
|
|
pH
|
7.351–7.463
|
7.310–7.462
|
7.351–7.443
|
7.277–7.409
|
7.34–7.38
|
|
PCO2
|
37 (31–43)
|
31 (25–37)
|
42 (38–46)
|
38.7 (33–45)
|
43 (40–46)
|
|
PO2
|
92 (81–103)
|
107 (95–118)
|
55 (45–65)
|
|
50–67
|
|
HCO3–
|
22 (19–25)
|
18 (14–22)
|
22.5 (20–24)
|
20.6 (18–23)
|
23.0 (22–24.5)
|
Interpretation of Acid-Base Balance
Interpretation of acid-base balance includes identification of the primary problem (acidosis vs. alkalosis), calculation of compensation, identification of mixed problems and clinical interpretation. Other means of analysis are calculation of the anion gap and Stuart's approach to acid-base disorders.
Acid-base disorders have an underlying primary problem. As the animal tries to maintain a physiological pH, compensatory changes will occur that are typical for each underlying problem (Figure 2).
The following five steps will allow interpretation of the primary problem and identification of mixed problems.
 pH change?
pH change?
 2 > 7.4
2 > 7.4  alkalosis
alkalosis
 2 < 7.4
2 < 7.4  acidosis
acidosis
 PCO2 or HCO3 change:
PCO2 or HCO3 change:
 Same direction as pH
Same direction as pH  metabolic
metabolic
 Opposite direction to pH
Opposite direction to pH  respiratory
respiratory
 Adequate compensation? Use Figure 3 to calculate expected compensatory changes in PCO2 (for metabolic acidosis/alkalosis) or HCO3 (for respiratory acidosis/alkalosis).
Adequate compensation? Use Figure 3 to calculate expected compensatory changes in PCO2 (for metabolic acidosis/alkalosis) or HCO3 (for respiratory acidosis/alkalosis).
 Identification of a mixed problem. The following findings are associated with a mixed problem:
Identification of a mixed problem. The following findings are associated with a mixed problem:
 Normal pH despite abnormal PCO2 or HCO3
Normal pH despite abnormal PCO2 or HCO3
 Overcompensation
Overcompensation
 Insufficient compensation
Insufficient compensation
 Anion gap (AG). Calculation of AG allows identification of metabolic acidosis due to unmeasured anions.
Anion gap (AG). Calculation of AG allows identification of metabolic acidosis due to unmeasured anions.
 AG = Na - Cl - HCO3 Normal < 20 (dog), < 27 (cat)
AG = Na - Cl - HCO3 Normal < 20 (dog), < 27 (cat)
 Increased AG: metabolic AG acidosis (methanol, uraemia, diabetes/diabetic ketoacidosis (DKA), paraldehyde, isopropyl alcohol, lactate, ethylene glycol, rhabdomyolysis, salicilate (aspirin)
Increased AG: metabolic AG acidosis (methanol, uraemia, diabetes/diabetic ketoacidosis (DKA), paraldehyde, isopropyl alcohol, lactate, ethylene glycol, rhabdomyolysis, salicilate (aspirin)  MUD PILERS).
MUD PILERS).
 Low AG: hypoalbuminaemia
Low AG: hypoalbuminaemia
Once acid-base disorders are identified, the findings should be evaluated in conjunction with clinical signs and differentials of the identified problem.
Figure 2. Primary acid-base problems with compensatory changes.
|
Primary problem
|
PH
|
PCO2
|
HCO3
|
|
Metabolic acidosis
|
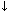
|
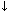
|
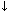
|
|
Metabolic alkalosis
|
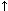
|
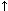
|
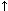
|
|
Respiratory acidosis
|
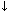
|
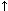
|
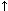
|
|
Respiratory alkalosis
|
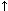
|
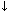
|
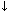
|
Figure 3. Expected compensatory changes in PCO2 (for metabolic acidosis/alkalosis) or HCO3 (for respiratory acidosis/ alkalosis).
|
Primary problem
|
|
Expected compensation
|
|
Metabolic acidosis
|
|
per  1 mmol HCO3- 1 mmol HCO3-
|
 0.7 mm Hg CO2 0.7 mm Hg CO2
|
|
Metabolic alkalosis
|
|
per  1 mmol HCO3- 1 mmol HCO3-
|
 0.7 mm Hg CO2 0.7 mm Hg CO2
|
|
Respiratory acidosis
|
Acute
Chronic
|
per 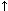 1 mmHg CO2 1 mmHg CO2
per 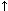 1 mmHg CO2 1 mmHg CO2
|
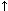 0.15 mmol HCO3- 0.15 mmol HCO3-
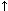 0.35 mmol HCO3- 0.35 mmol HCO3-
|
|
Respiratory alkalosis
|
Acute
Chronic
|
per 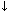 1 mmHg CO2 1 mmHg CO2
per 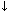 1 mmHg CO2 1 mmHg CO2
|
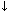 0.25 mmol HCO3- 0.25 mmol HCO3-
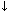 0.55 mmol HCO3- 0.55 mmol HCO3-
|
Differentials
Hyperchloraemic Acidosis
 Diarrhoea
Diarrhoea
 Renal tubular acidosis
Renal tubular acidosis
 Carbonic anhydrase inhibitors
Carbonic anhydrase inhibitors
 Dilutional acidosis (NaCl)
Dilutional acidosis (NaCl)
 Addisons disease, aldosterone deficiency
Addisons disease, aldosterone deficiency
Respiratory Acidosis
Hypoventilation:
 Upper airway stenosis
Upper airway stenosis
 Central respiratory depression
Central respiratory depression
 Cardiopulmonary arrest
Cardiopulmonary arrest
 Neuromuscular problem
Neuromuscular problem
 Severe pulmonary abnormalities
Severe pulmonary abnormalities
Metabolic Alkalosis
 Loss of Cl-: vomiting (gastric fluid), diuresis.
Loss of Cl-: vomiting (gastric fluid), diuresis.
 Mineralocorticoid excess: Cushing's, hyperaldosteronism.
Mineralocorticoid excess: Cushing's, hyperaldosteronism.
 Alkali administration: bicarbonate, phosphorus binder.
Alkali administration: bicarbonate, phosphorus binder.
 Others, e.g., refeeding syndrome, severe K or Mg deficiency
Others, e.g., refeeding syndrome, severe K or Mg deficiency
Respiratory Alkalosis
Hyperventilation:
 Hypoxaemia (anaemia, shock, V/Q mismatch, diffusion problem, shunt)
Hypoxaemia (anaemia, shock, V/Q mismatch, diffusion problem, shunt)
 Primary lung disease (pulmonary receptor stimulation): pneumonia, embolism, oedema
Primary lung disease (pulmonary receptor stimulation): pneumonia, embolism, oedema
 Hyperthermia (fever, heat stroke)
Hyperthermia (fever, heat stroke)
 Pain
Pain
Therapy
Most metabolic derangements can be corrected by appropriate fluid therapy and will not require drug therapy. Sodium bicarbonate may be indicated in patients with metabolic acidosis associated with renal insufficiency or severe metabolic acidosis despite appropriate fluid therapy (for example patient with DKA). Sodium bicarbonate is contraindicated in patients with respiratory acidosis as it will add to CO2 concentrations.
Respiratory acid-base abnormalities are treated by eliminating the underlying disease and appropriate ventilation therapy if required.
Interpretation of Oxygenation/ Ventilation
Oxygenation and ventilation are assessed using the following steps.
Oxygenation (PaO2)
 Normal PaO2 breathing room air is 80–100 mmHg
Normal PaO2 breathing room air is 80–100 mmHg
 With oxygen supplementation, normal PaO2 should be 4–5 times FiO2
With oxygen supplementation, normal PaO2 should be 4–5 times FiO2
Differentials for  PaO2 include:
PaO2 include:

 FiO2 (altitude)
FiO2 (altitude)
 Hypoventilation
Hypoventilation
 V/Q mismatch
V/Q mismatch
 Pulmonary diffusion problem
Pulmonary diffusion problem
 Cardiogenic: Right-to-left shunt
Cardiogenic: Right-to-left shunt
PCO2
 Increased
Increased  hypoventilation
hypoventilation
 Decreased
Decreased  hyperventilation (see above for differentials)
hyperventilation (see above for differentials)
Signs of V/Q Mismatch or Diffusion Problems
The following equations allow interpretation of pulmonary gas exchange:
 A-a Difference: PAO2–PaO2
A-a Difference: PAO2–PaO2
 PAO2 = FiO2 x (barometric pressure - 47) - 1.2 x PaCO2
PAO2 = FiO2 x (barometric pressure - 47) - 1.2 x PaCO2
 Normal: 0–15 with room air, < 100 with 100% O2
Normal: 0–15 with room air, < 100 with 100% O2
 PaCO2 + PaO2 should be < 120–160 mmHg at room Air
PaCO2 + PaO2 should be < 120–160 mmHg at room Air
 PaO2 / FiO2:
PaO2 / FiO2:
 Normal > 400
Normal > 400
 < 300: Acute lung injury
< 300: Acute lung injury
 < 200: Acute respiratory distress syndrome (ARDS)
< 200: Acute respiratory distress syndrome (ARDS)
Therapy
Insufficient oxygenation is treated by eliminating the underlying disease, oxygen therapy and mechanical ventilation in patients where oxygen therapy is not sufficient or increased work of breathing occurs with severely increased respiratory effort. Ventilation problems are treated by eliminating the underlying disease process (analgesia, thoracocentesis, etc.), and mechanical ventilation in patients with hypoventilation.