C.A. Tony Buffington, DVM, PhD, DACVN
INTRODUCTION
Urinary tract stones are a relatively common problem in dogs and cats. Most stones in dogs and cats have one major crystal component, which are presented in Table 1.
Urine is a complex solution containing many organic and inorganic ions. More of these ions can remain in solution in urine than in water because of the complex interactions occur among the various organic and inorganic constituents in urine. For several possible reasons (e.g., diet, decreased water intake, altered urine pH, relative lack of inhibitors of crystallization), the solubility product of a particular crystal may be exceeded, crystals may form, and these crystals may aggregate and grow (see Figure).
Table 1
|
Stone type |
Dogs |
Cats |
|
Struvite |
55% |
~45% |
|
Calcium oxalate |
27% |
~45% |
|
Urate |
7% |
7% |
|
Cystine |
1% |
<1% |
|
Silicate |
1% |
0% |
Factors involved in the development of urolithiasis
Because urine is commonly supersaturated, observation of crystals in the urine does not mean the patient is at risk for urolithiasis. Supersaturation of urine depends on the amount of the ion ingested and excreted in the volume of urine produced. For this reason, reducing the ion concentration and increasing the urine volume and frequency of urination are the most important factors controlling urinary stone formation.
These relationships are shown in Figure 1.
Figure 1. Factors involved in the growth of crystals in urine. From: Drach GW: Urinary lithiasis. In: Campbell's Urology (ed 4) Harrison JH et al. (eds) WB Saunders, Philadelphia, 1978, p 793.
|
|
UNSTABLE: OVERSATURATED
--rapid growth
--aggregation of crystals
--spontaneous nucleation |
|
|
|
________________________ |
FORMATION PRODUCT |
|
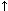 Increasing concentration Increasing concentration
of crystallizable substance
in urine
|
METASTABLE: SUPERSATURATED
--growth from previous crystals
--aggregation of crystals
--dissolution unlikely |
|
|
|
________________________ |
SOLUBILITY PRODUCT |
|
|
STABLE: UNDERSATURATED
--no nucleation or growth
--dissolution |
|
Urine pH also affects crystal formation, as shown in Table 2. The urine pH can be affected by diet, intrinsic factors (renal tubular acidosis), and external stressors (hyperventilation syndrome).
Table 2
|
More soluble in acidic urine (pH <~6.8) |
Struvite, calcium carbonate, calcium phosphate, urate |
|
More soluble in alkaline urine (pH >~7.0) |
Uric acid, cystine |
|
Minimal effect of urine pH |
calcium oxalate |
Conditions and drugs (e.g., anticholinergic agents) that predispose to urine stasis also play an important role in stone formation, because crystals must reside in the urinary tract for a sufficient time for a urolith to form.
Struvite
The composition of struvite stones is MgNH4PO4.6H20. Calcium phosphate (as carbonate apatite) often is present in small amounts (2-10%). The presence of three cations--Ca+2, Mg+2,and NH4+--detected by earlier qualitative methods was responsible for the name "triple phosphate" previously used for these stones. Struvite stones are spherical, ellipsoidal, or tetrahedral in shape and may be present singly or in large numbers of varying sizes. In dogs and cats, the bladder is the most common site of struvite stone formation, although they may occur at any site in the urinary tract. In dogs, struvite calculi tend to recur after surgical removal the recurrence rate in one study was 21%. In dogs, urinary tract infection by urease-positive bacteria (especially Staphylococci and Proteus sp.) plays the most important role in struvite stone formation. Hydrolysis of urea by these urease-positive bacteria liberates ammonia and carbon dioxide, which increases the pH of the urine and the availability of ammonium and phosphate ions for struvite formation. The solubility of struvite is markedly reduced in alkaline urine due to removal of protons from phosphate ions. Struvite solubility is reduced in animals with persistently alkaline urine even in the absence of urinary tract infection. In dogs that form struvite stones in the absence of urinary tract infection, predisposing factors include a family history of struvite stones, a diet based on vegetable proteins, and distal renal tubular acidosis. Urinary tract infection usually is NOT present in cats with struvite stones.
Oxalate
Calcium oxalate is the most common (kidney) stone type in people, and their incidence has been increasing in dogs and cats during the past 15 years. These stones are composed of calcium oxalate monohydrate (whewellite) or calcium oxalate dihydrate (weddellite). Oxalate frequently is not detected by qualitative analysis, and quantitative analysis is necessary. Calcium oxalate calculi usually are white in color and very hard, they often have sharp, jagged edges and may be single or multiple in number. They are found most often in the bladder and urethra; their recurrence rate may be as high as 25-48%. Urinary tract infection, when it occurs, is thought to be a complicating rather than a predisposing factor to oxalate urolithiasis.
Altered calcium metabolism also may play a role in development of oxalate urolithiasis. Increased urinary excretion of calcium (hypercalciuria) can result from increased absorption of calcium from the intestinal tract ("absorptive" hypercalciuria), from increased urinary loss of calcium ("renal leak" hypercalciuria), or from increased release of calcium from bone ("resorptive" hypercalciuria). In "absorptive" as compared to "renal leak" hypercalciuria, urinary calcium excretion is higher after feeding than during fasting. Chronic acidosis may be associated with increased urinary excretion of calcium due to increased calcium release from bone. Long term feeding of an acidifying diet may contribute to this "resorptive" hypercalciuria.
In one study, Miniature Schnauzers had higher urinary calcium excretion during fasting than did Beagles and urinary calcium excretion increased 3-fold after feeding (i.e., hypercalciuria seemed to be "absorptive"). Dogs with hypercalcemia due to primary hyperparathyroidism may develop calcium oxalate (or calcium phosphate) stones due to parathyroid hormone-mediated mobilization of calcium from bone ("resorptive" hypercalciuria).
Citrate forms a soluble complex with calcium and normally may be an inhibitor of calcium oxalate formation. Acidosis may be associated with decreased urinary citrate excretion and thus may predispose to calcium oxalate stone formation.
Urate
Urate stones in dogs are composed of the monobasic ammonium salt of uric acid (ammonium acid urate) Calcium oxalate may be a secondary component of some urate stones, and urate stones found in dogs with portosystemic shunts often contain struvite in addition to urate. Urate stones are found most often in the Dalmatian and English bulldog breeds, but other breeds also may be affected. Urate stones may be found in dogs with portosystemic shunts due possibly to reduced conversion of ammonia to urea and uric acid to allantoin. A defect in uric acid metabolism in some Dalmatian dogs predisposes them to urate stone formation. This defect is merely a predisposing factor and not a primary cause of urolithiasis because Dalmatian dogs that do not develop stones excrete as much urate as stone-forming Dalmatian dogs do, and because other breeds (e.g., English bulldog) also may develop urate urolithiasis.
Treatment
General
Relief of urinary tract obstruction and reestablishment of urine flow, and correction of fluid, electrolyte, and acid-base imbalances associated with obstruction and post-renal azotemia are the first steps in management of urolithiasis. A stone should be retrieved and submitted for analysis whenever possible. All dogs with urolithiasis should have their urine cultured. If urinary tract infection is present, appropriate antibiotic therapy and careful follow-up should be instituted to ensure elimination of infection.
The primary therapy for urinary stone disease is to reduce the urine specific gravity and increase the frequency of urination. Patients that have formed a stone should never be fed food dry. This is not to say that they cannot be fed dry food, just that at least a cup of water should be added to each cup of dry food before feeding so that enough time can pass for the food to completely absorb the moisture. Patients also may be fed canned formulations, or water, other liquids, or salts may be added to the diet. The aim is to reduce the urine specific gravity to < 1.020, or to double urine output. Patients must be allowed frequent opportunities to void to prevent bladder distension. Salt should not be given to patients at risk of fluid retention, nor for dogs with oxalate and cystine urolithiasis because natriuresis may cause hypercalciuria and may increase urinary excretion of cystine. We recommend to clients that the urine of their stone-forming pets be consistently clear, colorless, odorless, and often.
Because of the primary role of urinary tract infection by urease-positive organisms in struvite urolithiasis of dogs, careful elimination of infection by appropriate antibiotic therapy and repeated patient follow-up to demonstrate eradication of infection are the most important aspects of medical management to prevent recurrence.
Struvite
The use of urinary acidifiers to maintain urine pH in the range of 6.0-6.5 has been suggested in dogs because struvite and hydroxyapatite are most soluble in acidic urine. In most dogs with struvite urolithiasis, eradication of urinary tract infection will return urine pH to the acidic range. Use of urinary acidifiers in the presence of infection by a urease-positive organism is futile. If urine pH remains alkaline after elimination of urinary tract infection, other potential causes (e.g., dietary, familial, metabolic) of alkaline urine should be investigated. In cats with struvite urolithiasis without urinary tract infection, urinary acidifiers played a more important role, until most commercial cat foods were reformulated to reduce urine pH. Urine acidifiers should only be given to cats with urine pH > 6.5 measured at home under ad libitum feeding conditions, since travel to a veterinarian can result in an alkaline urine due to hyperventilation. There is some concern that addition of acidifying compounds to cat foods may have contributed to the increasing incidence of calcium oxalate stones in cats. It seems more likely that it unmasked a population of susceptible individuals.
A calculolytic diet (s/d®, Hill's Pet Nutrition, Inc.) has been used successfully to induce dissolution of naturally occurring struvite calculi in dogs and cats. In dogs, the diet promotes under saturation of the urine with ions necessary for formation of struvite uroliths and thus promotes dissolution of existing struvite calculi. Clinical findings in patients fed s/d® include polyuria/polydipsia and dilute urine, decreased BUN, increased alkaline phosphatase (hepatic isoenzyme), decreased serum phosphorus, and decreased serum albumin concentrations. In dogs with struvite uroliths and urinary tract infection, dissolution is expected to take 2-3 months. The diet is used for one month beyond radiographic evidence of urolith dissolution.
Certain precautions should be observed when considering use of s/d® diet. The canine form should not be fed to growing puppies, pregnant or lactating bitches, or cats because of its extremely low protein content. Occasionally, nephroliths that have decreased in size after institution of the diet may pass into the ureter causing ureteral obstruction and hydronephrosis. No comparisons between this and other recently introduced diets designed for struvite dissolution are available.
Oxalate
Attempts to dissolve calcium oxalate stones in dogs and cats have so far been unsuccessful and surgery is required to remove stones. Post-operatively, a diet low in calcium and oxalate should be fed. Dietary phosphorus should not be restricted because reduced phosphorus could result in increased activation of vitamin D3 to calcitriol by 1-α-hydroxylase in the kidney and cause increased intestinal absorption of calcium. Also, urinary pyrophosphate may function as an inhibitor of calcium oxalate formation. Dietary magnesium should not be restricted because it may serve as an inhibitor of calcium oxalate formation. The diet should not be supplemented with sodium because natriuresis is associated with hypercalciuria. A number of commercial diets recently have become available that attempt to prevent recurrence of oxalate stones. Only the feeding of the Waltham S/O diet has been shown to result in urine that is under saturated for both struvite and calcium oxalate.
Urate
Dissolution and prevention protocols include some combination of urine dilution, low purine diet, alkalinization of urine, and allopurinol. Diets low in organ-derived meats reduce the ingested purine load. Feeding a low protein, low purine diet has been shown to reduce urinary excretion of urate in normal dogs. A purine-restricted, non-acidifying diet (e.g., Hill's u/d®) has been recommended for dogs with urate urolithiasis, but no studies are available confirming its efficacy. If the dry form of the diet is fed, at least one cup of water per cup food should be thoroughly mixed with the diet before feeding. If stones recur, allopurinol is a competitive inhibitor of the enzyme xanthine oxidase, which converts hypoxanthine to xanthine and xanthine to uric acid in the course of purine metabolism may be considered. It is recommended for use in dogs with urate urolithiasis at a dosage of 30 mg/kg/day divided BID. A dosage of 30 mg/kg/day divided BID for one month, followed by 7-10 mg/kg/day has been recommended for prevention of recurrence. The dosage of allopurinol should be reduced in the presence of renal failure since the kidneys excrete it.
More information is available at: http://www.nssvet.org/